Warning: Trying to access array offset on value of type null in /home/elisew5/public_html/drpeterwitt.com/wp-content/plugins/contextual-related-posts/includes/content.php on line 49
Images Collection
View this article in Search Friendly Plain Text
NOTE: This plain text article interpretation has been digitally created by OCR software to estimate the article text, to help both users and search engines find relevant article content. To read the actual article text, view or download the PDF above.
Holden, W. 1977. Behavioral evidence of chemoreception on the legs of the spider Araneut
a. J. Arachnol. 3:207-210.
BEHAVIORAL EVIDENCE OF CHEMORECEPTION ON THE LEGS
OF THE SPIDER ARANEUS DIADEMA TUS CL.
THE JOURNAL OF ARACHNOLOGY
Wendy Holden
North Carolina Mental Health Services
Research Section
P.O. Box 7532
Raleigh, North Carolina 27611
ABSTRACT
Behavioral responses of the spider Araneus diadematus to chemically-treated prey support the
hypothesis that hairs on the legs of spiders receive chemical stimuli. Analysis of movie films showed a
definite withdrawal reaction of spiders to quinine-coated flies and repeated trials with quinine-treated
flies led to significantly (P <^0.01) longer periods of time for the spiders to respond to prey.
The frame by frame analysis of movies has provided the evidence needed to conclude
that spiders are capable of receiving sufficient signals to stimulate the chemosensitive
hairs on the legs. While conducting an experiment on altering the behavioral pattern in
the feeding of the spider, it was perceived that, when it was exposed to prey coated with
a bitter solution, the animal did not have to touch the prey with its mouth parts to be
repelled. Upon observing this, I thought a closer observation was necessary. A movie
could freeze each action of the spider so that any movement which occurred, that was
too fast for the naked eye, could easily be analyzed frame by frame. After a close study
of the movie it was determined that the spider did not have to touch a quinine solution
with its mouth parts at any time, in order to be repelled, even if the coated fly was
moving about and vibrating.
Several hairs on the spiders’ legs are morphologically similar to the hairs on the
proboscis of the blowfly (Foelix, 1970). The blowfly has been studied morphologically
and electrophysiologically by Dethier (1955, 1971) and Wolbarsht (1958) who estab-
lished that the four chemoreceptors on the proboscis have many functions dealing with
sensing, coding and transmitting. In the spider, chemoreceptor structures have been des-
cribed but, “… there was never any supporting evidence” for the function (Foelix,
1970). McCook (1890) wrote “I have long entertained the opinion that the sense of smell
in spiders abides entirely in the delicate hairs.” Bays (1962) provided sufficient evidence
that spiders could learn to distinguish between two vibrations and two different tastes:
glucose (sweet) and quinine (bitter). The spiders in Bays’ experiments, after a few trials,
rejected the quinine-coated prey, which leads one to believe that for the spider there must
be some aversive properties of the bitter solution.
Chemicals contained in the fresh silk or on a mature female spider appear to play a
vital role in the pre-mating process. According to Kaston (1936) the male of Dolomedes
scriptus, a non-orb-weaver, does not attempt to court if the female’s leg has been dipped
into ether, while courtship is elicited by the ether extract, after evaporation of the ether.
208
THE JOURNAL OF ARACHNOLOGY
Wolff and Hempel (1951) find their spiders sensitive to chemical stimuli on touch, and
Krafft (1971) interprets his observations of social spiders’ interaction as transmitted
through chemical and tactile signals.
There can be no doubt that spiders perceive non-volatile chemicals on contact (Kaston,
1936); but is a touch by the leg sufficient to receive the chemical indicating the taste of
food?
METHOD
Twenty-four Araneus diadematus spiders were used in the experiment, 12 in each
group, the experimental and control groups were kept on different diets. The laboratory
conditions were controlled and the temperature was regulated (cool and dark from mid-
night until 8:30 a.m. and light and warm until midnight). The animals were kept in cages
with screen on the sides and with glass in the front and back (for details see Witt, 1971).
The experimental group was fed a suspension of 30 to 40 homogenized fly abdomens in
approximately two teaspoons of water with a small quantity of sugar. About 0.01 ml was
put into a syringe which was held to the mouth parts of the spiders until imbibed by the
animal five times a week for 47 days. The control group received two untreated flies a
week for 47 days.
The fly for the experimental spiders was first fully dipped into a suspension of quinine
in water, then put onto the web. Usually the fly was wiggling when it was placed onto the
web and this attracted the spider’s attention. The spider approached the fly, touched it
with a front leg or put all of its legs on the prey, then either went back up to the hub
(center) of the web or wrapped it first and then went up. This sequence of events would
follow the same pattern each time a quinine-covered fly was used. If the prey was not
wiggling when it was put onto the web, the tuning fork (middle C) was held just below
the prey to lure the spider. In some cases, an uncoated fly was held with forceps just
below the quinine coated prey until the spider approached. The quinine coated prey was
given each day on which the spider had built a new web.
RESULTS AND CONCLUSIONS
This experiment was originally performed as a followup study to experiments by
Reed, et al (1970) which indicated that while experience did not change web-building
behavior, it influenced the spiders’ handling of prey. The experimental spiders were
“timed” after a period of 47 days, during which they were given the quinine coated prey,
and compared to the control group’s timing. With the aid of a stop-watch I ascertained
the interval between the time when the prey was touched to the web and when the spider
touched the prey. After several trials the spiders in the experimental group took signifi-
cantly (P<0.01) longer periods of time to get to the prey than did the control group (see
Table 1): their feeding behavior was changed as a consequence of experience.
Quinine solution has no odor, so there is no airborne stimulus. It is true that the spider
could detect the quinine-coated fly was wet to the touch; but how did the spider know
that the fly had not been dipped into a glucose solution, which it usually takes without
hesitation (Bays, 1962)? There was a definite withdrawal reaction from the quinine,
indicating that the spider perceived an unpleasant stimulus through sense organs on the
tarsi (see Figs. 1, 2). Combining these observations with those of Foelix (1974) it seems
HOLDEN-CHEMORECEPTION IN ARANEUS DIADEMATUS
209
safe to suggest that the spider received sufficient chemical stimuli with the chemosensitive
hairs on the tarsus to recognize the substance. Such a hypothesis is supported through the
frame by frame observation of the spider’s movements in a movie film, as the animal
approaches the quinine covered prey, and responds.
The author gratefully acknowledges the support of the National Science Foundation
Grant No. GB-15174 to Dr. Peter N. Witt.
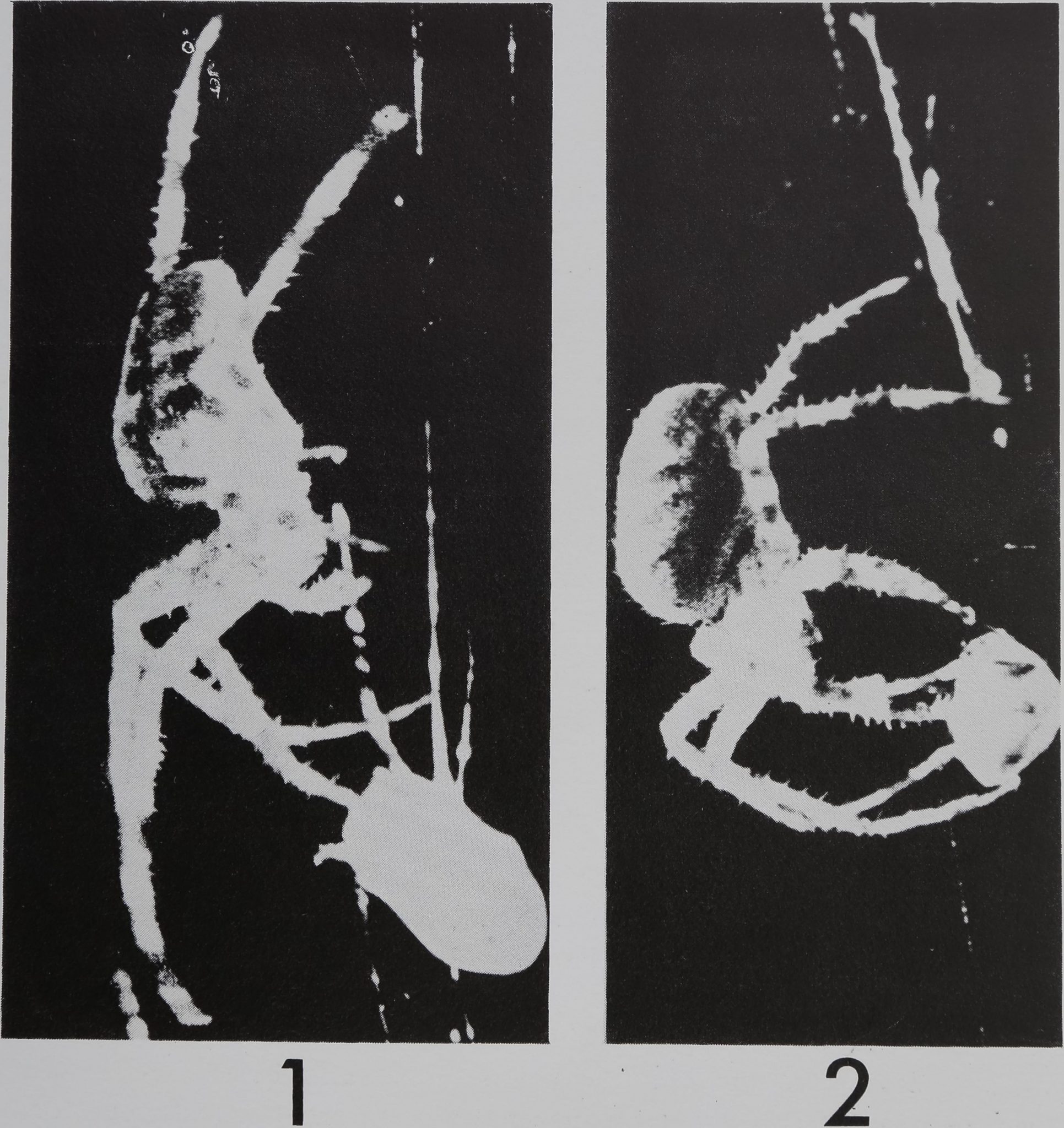
Fig. l.-This is an enlarged picture taken from one fr^une of a 16 mm movie.’It shows a female
Araneus diadematus spider (on left) at its closest distance to a fly coated with quinine (below on the
right). The frames before and after this picture reveal the spider at a farther distance from the fly.
(Notice that the bitter substance is touched by the tarsus only, before the spider retreats.)
Fig. 2.-This shows a different Araneus diadematus female spider under similar circumstances as in
Fig. 1. Notice the spider (on the left) touches the prey (on the right) with the tips of all the legs
except the back ones which hold on to the web. In this sequence, the mouth did not come in contact
with the prey at any time.
210
THE JOURNAL OF ARACHNOLOGY
Table l.-Mean and standard deviation of time it took female Araneus diadematus littermates to
reach prey at 1.4 cm distance, after the experimental group had been offered quinine-coated flies
under similar circumstances for 47 days, five times a week, and had been fed spider suspension from a
syringe held to the mouth, while controls caught flies in webs. The difference in time is significant
below the 1 per cent level.
Number of Mean time Standard
Animals for prey in seconds Deviation
Control Group 2ll 9.66 ± 9.25
Experimental Group 6 38.16 if36-22
LITERATURE CITED
Bays, A. M. 1962. A study of the training possibilities of Araneus diadematus C1. Experientia (Basel)
18:423.
Dethier, V. G. 1955. The physiology and histology of the contact chemoreceptors of the blowfly.
Quart. Rev. Biol. 30:348-371.
Dethier, V. G. 1971. A surfeit of stimuli: a paucity of receptors. Amer. Sci. 59:706-715.
Foelix, R. F. 1970. Chemosensitive hairs in spiders. J. Morphol. 132:313-334.
Kaston, B. J. 1936. The senses involved in the courtship of some vagabond spiders. Entomol. Amer.
16:97-167.
Krafft, B. 1971. La société d’Agelena consociata Denis, araignée sociale du Gabon. 96 Congr. Nat. Soc.
Sav. B:453.
McCook, H. A. 1890. American spiders and their spinning work, Philadelphia, 2:299.
Reed, C. F., P. N. Witt, M. B. Scarboro and D. B. Peakall. 1970. Experience and the orb web.
Developm. Psychobiol. 3:251-265.
Witt, P. N. 1971. Instructions for working with web-building spiders in the laboratory. BioScience
21:23-25.
Wolbarsht, M. L. 1958. Electrical activity in the chemoreceptors of the blowfly. II. Responses to
electrical stimulation. J. Gen. Physiol. 42:413-428.
Wolff, D. and U. HempelM951. Versuche uber die Beeinflussung des Netzbàues von Zilla-x-notata
durch Pervitin, Scopolamin und Strychnin. Z. vergl. Physiol. 33:497-528.