Warning: Trying to access array offset on value of type null in /home/elisew5/public_html/drpeterwitt.com/wp-content/plugins/contextual-related-posts/includes/content.php on line 49
Images Collection
View this article in Search Friendly Plain Text
NOTE: This plain text article interpretation has been digitally created by OCR software to estimate the article text, to help both users and search engines find relevant article content. To read the actual article text, view or download the PDF above.
Revue Arachnologique, 2 (3)t 1979: 103-132.
Comparative studies of Dictyna and Mallos
(Araneae, Dictynidae)
II. The relationship between courtship, mating,
agression and cannibalism in species with differing types
of social organization
R.R. Jackson*
(North Carolina Mental Health Research,
P.O. Box 7532, Raleigh, North Carolina 27611)
– I.
Contents
Résumé, summary. . . . . . . . . – . . . . „ . . . . . . . . 104
Introduction. . . . . . . . . . . . ” ; . . ; . . . …. 104
‘ Methods and materials.. …………………. . . . . . . . . . … . 107
Results. . . . . . . . . . i . . . . . …. . . . . . . 107
L Elements of behavior. … . . . . . . . . . . ….. 107
II. Organization of behavior during interactions in the laboratory. ,. . . . 113
m. Mating in the laboratory. . . . . . . . . . . . , . .. . … 115
IV. Observations from the natural habitats of the spiders. . . •. . . . . 117
V. Spinning behavior during courtship. . …… … … 117
. VI. The role of sexual dimorphism in communication. . . . . … . 117
VII. Aggression. ………………………………………………… 121
Vni. Spacing tendencies. ……………………………………….. 123
IX. Cannibalism. …………………………………………………124
Discussion……………….. . . . …………………..125
I. Interspecific differences in courtship. . ‘ . 1………………125
II. Elements of behavior in other species……………………….. 126
III. The non-cannibalistic nature of Mallos gregalis. ………………..126
IV. The non-aggressive nature of Mallos gregalis ‘ .127
V. The function of courtship. ………………………………. . 127
Acknowledgements………………………………………………… 129
References………………………………………………………… 129
* Manuscrit reçu le 10 mai 1978; adresse actuelle de : Zoology Department, University of
Canterbury, Christchurch 1, New Zealand.
104
R.R. Jackson
Résumé
L’organisation sodale chez les Dicîyna et les Mallos comprend des espèces solitaires,
des espèces qui vivent en groupes et gardent des territoires (grégaires, territoriales), et une
espèce vivant en groupe (Mallos gregalis) qui ne conserve pas de territoires (grégaire, non-
territoriale). Dans toutes les espèces, les mâles semblent être plus nomades que les fe-
melles; les femelles, plus sédentaires. Les femelles s’accouplent avec plusieurs males. Les
dimorphismes sexuels des chélioères et des palpes pourraient être importants quant à leurs
moyens de communiquer entre eux. Par contraste avec les autres espèces, le ML gregalis
n’est pas agressif, il vit à proximité de ses voisins et n’est pas cannibale. La façon de
faire la cour et l’accouplement varient selon les espèces et à l’intérieur même de ces
dernières; cependant la façon de courtiser chez les espèces agressives et cannibales ne
semble ni plus complexe ni plus circonspecte que celle des M. gregalis. Ces observations
jettent un doûte sur l’hypothèse avancée précédemment que la principale fonction de
faire la cour chez les araignées est de protéger les mâles contre les femelles cannibales.
Summary
Social organization within Dictym and Mallos includes solitary spedes, species that
live in groups and maintain territories (communal, territorial), and a group-living spedes
(Mallos gregalis) that does not maintain territories (communal, non-territorial). In all
spedes, males seem to be more nomadic; females, more sedentary. Females will mate with
more than one male. Sexual dimorphisms of the chelicerae and pedipalps may be involved
in communication. In contrast to other species,M. gregalis is non-aggressive, close-spacing,
and non-cannibalistic. Courtship and mating behavior vary within and among spedes;
however, the courtship of aggressive and cannibalistic spedes seems no more complex or
cautious than that of M. gregalis. These observations bring into question the previously
proposed hypothesis that a major function of courtship in spiders is to protect males from
cannibalistic females. / V \ V-
Introduction
In spiders, cannibalism has repeatedly been given major importance, either expli-
dtly or implicitly, in discussions concerning the function of courtship (e.g., BRISTOWE,
1958; BRISTOWE & LOCKET, 1926; GERHARDT & KAESTNER, 1937; KRAFFT,
1970; PLATNICK, 1971; SAVORY, 1928; TURNBULL, 1973; WITT, 1975). Spiders
aie predators of arthropods, and the males of most species are within the size range of
the prey of females. Observations in nature and espedally in the laboratory indicate that
female spiders sometimes kill and feed on conspedfic males. Often the courtship behavior
of males has a “cautious” appearance, with periods of approaching and withdrawing;
and the female may rush toward the male in a manner appearing rather “violent”. Obser-
Courtship and Dictynidae social organization
105
valions such these might seem to compel the conclusion that spider courtship behavior
is largely the result of natural selection related to cannibalism. In other words, the func-
tion of courtship tends to be viewed as reducing the probability that the male will be
treated as prey by the female. This will be called the “cannibalism reduction hypothesis”
for the fonction of male courtship. Although the views of some authors tend to be
complex, allowing for other functions in addition to cannibalism reduction, some varia-
tion of the cannibalism reduction hypothesis is a nearly ubiquitous element in discussions
of spider courtship. Sometimes it is an explicitly proposed function; other times it is
merely implied.
The popular myth that female spiders usually prey on the males either during court-
ship or after copulation has been disclaimed many times by arachnologists. Sometimes
the same arachnologists emphasize cannibalism reduction in discussions of the function
of courtship. Perhaps cannibalism is not so frequent in spiders because of the effectiveness
of courtship in preventing its occurrence, but this hypothesis needs critical investigation.
Some observations on the behavior of dictynid spiders that are relevant to the
cannibalism reduction hypothesis will be discussed here. These spiders are of special
interest because different species live under differing types of social organization (JACK-
SON, 1978a), and the cannibalistic tendencies of the species vary with their social orga-
nization. The majority of dictynids are solitary, each individual generally living alone in
an individual web that does not touch other occupied webs. These are found especially
on stems and leaves of shrubs and herbaceous plants. Mallos trivittatus Banks, Dictyna
calcarata Banks, and D. albopUosa FranganiDo are communal and territorial, living in web
complexes that consist of individual web units connected to each other by silk. Although
several individuals often occupy a single web unit, generally two adults of the same sex
or two immatures of comparable size do not share the same web unit. Each individual
web and each web unit consists of a catching area and a nest in which the spiders tend
to remain when not active or feeding. Mallos gregalis Simon is communal and non-territo-
rial, with hundreds of individuals sharing the same large communal webs which are not
divided into web units. Females of M. trivittatus tend to be 7mm in body length; males
5mm. The other species in this study tend to be smaller; females approximately 5mm,
males usually I1™11 shorter.
Species with differing types of social organization will be compared with respect
to courtship, mating, aggression, and cannibalism. Courtship is defined as heterosexual
communicatory behavior that forms the normal preliminaries to mating (JACKSON,
1977a); mating is used as synonym for copulation. WlLSON*s (1975) definition of com-
munication will be used: “Action on the part of one organism (or cell) that alters the
probability pattern of behavior in another organism (or cell) in a fashion adaptive to
either or both participants.” Signals are behavioral and other characteristics of an orga-
nism used in communication (OTTE, 1974). Aggression is behavior of one individual that
reduces the freedom or fitness of another individual (WILSON, 1975), with usage restric-
ted to intraspecific interactions for the present discussion. Cannibalism is intraspecific
predation (FOX, 1975). Whether cannibalism is motivationally distinct from other forms
of aggression (see HUNTINGFORD, 1976;MOYER, 1968) has not been determined for
these spiders.
R.R. Jackson
Fig. 1. — Maintenance cage constructed from clear plastic petri dish (diameter: 9 cm).
Diameter of all holes: Ie”1. a: Hole plugged with cork, b: Hole covered by metal
screen, for ventilation, c: Cotton roll (4cmx Ie®) inserted through hole. Opposite
end set in glass jar (d) containing water, providing continual moisture to interior of
cage, e: Culture of Drosophila melanogaster in glass vial, f: Plastic cap with hole,
g: Plastic tube inserted at one end through hole in lid of culture vial and at other
end through hole in cage. Flies emerge from culture and travel through tube into
cage, providing continual food for spiders. Fresh culture vials substituted as neoes-
sary.
Courtship and Dictynidae social organization
107
Methods and materials
General methodology will be described here. Specific methods used in studies of
spacing and cannibalism will be given in the appropriate sections.
In the laboratory. At gregalis in large communal webs were maintained on a diet of
houseflies [Musca domestica), provided at approximately 5-day intervals. Temperature
was maintained at approximately 24°C, and the light cycle was approximately 13 L: 11 D.
There colonies were begun from spiders collected by Burgess (see BURGESS, 1976) in
Guadalajara, Mexico. The webs in the laboratory were on plants and other objects, and
they were not enclosed.
Small groups and single individuals of M. gregalis and single individuals of M, tri-
vittatus. At. niveus Oi\ Cambridge, and D. calcarata were also maintained in cages (fig. 1)
similar in design to ones that have been used for other types of spiders (JACKSON, 1974).
Since the dictynids tended to fasten their webs primarily to the corks and lower portion
of the cage, the lid could usually be removed without substantially damaging the web.
The usual diet wa$ Drosophila, although occasionally adult Musca domestica (houseflies)
were inserted through the cork holes as either supplementary or substitute prey. At. tri-
vittatus and At. niyeus were collected in Arizona and D. calcarata was collected in Jalisco,
Mexico.
To observe interactions, one spider was introduced into the cage of another on the
side most distant from the resident spider. A clear plastic tube (I011 in diameter, 3cm
long) was used for transferraL The spider was pushed into the tube with a cameFs hair
brush, and the two ends were stoppered with corks. Then the lid was removed from the
cage of the other spider, and the corks were removed from the tube. Holding the tube
over the web, a brush was inserted through one end, forcing the spider from the other
end and onto the web.
Whenever it was necessary to select spiders and assign them to groups, this was done
with a random numbers table (RÖHLF and SOKAL, 1969). The statistical tests used
are described by SOKAL and ROHLF (1969).
Results
I. Elements of behavior
The following elements of behavior were discerned from observing At. gregalis, At.
trivittatus, and D. calcarata in the laboratory.
1. Twich abdomen.
Abdomen twitching occurred intermittently in all three species during courtship,
copulation, and intersexual interactions. It occurred both concurrent with other activities
and while the spiders were otherwise inactive, and the details of this behavior differed
among the species.
Similar behavior has been observed in numerous spider species (GERHARDT &
KAESTNER, 1937), but its function is not clear in most cases. In a few species, this
behavior is associated with stridulation (GwinnER-Hanke, 1970; LEGENDRE, 1963)
108
R.R. Jackson
or sound production when the abdomen strikes the substrate (HARRISON, 1969), but
there is no evidence of either in the species considered here.
Abdomen twitching by males always occurred during courtship and sometimes
occurred during male-male interactions. Occasionally females of 1\t trivittatus and D.
calcarata twitched their abdomens when interacting with males. In At gregalis, abdomen
twitching tends to be relatively inconspicuous. The male’s abdomen moves at a high
frequency (estimated: 10 cycles per sec.) and low amplitude in the sagittal plane (“up
and down’*). In At trivittatus and D. calcarata, the amplitude is greater and the frequency
seems slower (estimated: 2 or 3 per sec.). In D. calcarata, movement is in the sagittal
plane, as in At gregalis. In At trivittatus, movement is in the frontal plane (“back and
forth”).
2. Charge.
Charging spiders ran toward other spiders, over a distance of 1 to 2cm, suddenly
stopping before contacting the other spider.
3. Chase.
During chasing, one spider followed another with both moving rapidly. The chasing
spider sometimes touched the chased spider with Its forelegs. The duration of individual
chases was usually approximately 1 sec. Females sometimes chased and charged towards
courting males, and these elements of behavior frequently occurred during male-male and
female-female Interactions also.
4. Pluck and tap silk.
A number of different motor patterns can be described as plucking and tapping on
the web. Although these movements were made primarily while the spiders were not in
physical contact, occasionally they occurred briefly while the spiders were touching.
Females, males, and immatures of each species tapped the web with their legs I during
normal locomotion, even when not in the presence of other individuals. This tapping has
a relatively smooth, slow appearance, as the spider waved its legs around, tapping the
web several times with one leg then the other. During interactions between spiders, males
and females of all species sometimes performed movements with some similarities to this
tapping pattern,but with a more jerky, sudden, and rapid time-course and appearance (fig.
2). Both tapping (tarsi strike silk as legs move ventrally) and plucking (daws hold thua
release silk as the legs move dorsally) were seen at least occasionally when the interacting
spiders were observed under a microscope. Behavior of this type was more common in
courtship interactions, but also occurred during male-male and female-female interactions.
5. Jerk.
While standing in one location. At trivittatus males sometimes suddenly and rapid-
ly flexed all of theirlegs. Flexion of leg I was the most conspicuous. Only one jerk occurred
at a time, followed by a pause of at least 1 sec. Occasionally males slowly walked forward
as they intermittently jerked.
6. Quiver*
Superimposed on jerking. At trivittatus sometimes undertook a series of very rapid,
low amjriitude movements of the body. (Jerking and quivering were seen during court-
ship only.)
Courtship and Dictynidae social organization
109
7. Rock.
During courtship, males of D. calcarata sometimes rhythmically moved their bodies
in the sagittal plane (“up and down”) in a relatively slow, smooth manner, by alternately
extending and flexing their legs.
8. Pluck walk.
This very distinctive behavior was performed by only the males of M. gregalis. It
seems to constitute not only a component of courtship but also serves as a broadcasted
sexual advertisement signal (JACKSON, 1978b). As males walked slowly through the
web, they plucked in a rather jerky and rapid manner, 1 to 7 times with one leg I and
then with the other. Each series of plucks with a given leg usually takes place in less than
1 sec. and the switch-over to the other leg occurred rapidly. If another spider was near, the
male would stop walking and pluck in this manner briefly while standing in one location.
Each leg I was held outward at approximately 45° to the sagittal plane during plucking.
While his claws hooked around the silk, the male moved his legs I posteriorly and medial-
ly to pluck.
9. Sprint.
At trivittatus mates sometimes approached females by alternately walking, pausing,
and sprinting. Sprinting was always preceded by a pause. Then the mate suddenly moved
forward very rapidly over a distance of a few mQimeters (sometimes taking only a single
step).
10. Inteiplay of legs.
When the legs of two spiders interplayed, primarily the tegs I were involved (fig. 3).
The tegs of the two spiders repeatedly touched and moved slowly. This behavior has not
been recorded in detail, but no particular pattern or stereotype was evident. Interplay of
legs was seen during every type of interaction of each species. In At gregalis this was
virtually the only type of behavior that was seen when femafes or immatures encountered
each other, and even this did not seem to occur in most cases, with the spiders simply
changing directions and walking away.
11. Stroke and tap with legs.
During courtship, females and especially males of all three species may stroke and
tap, with their legs I and sometimes with their tegs II, on the legs, céphalothorax, and
especially abdomen of the other spider. The form of these movements has not been recor-
ded in detail.
12. Touch with face.
A distinctive and frequent element in the courtship of all species observed was for
the male to bring his face (i.e.* anterior céphalothorax) into contact with some part of
the femaleVbody (fig. 4). Every copulation observed was preceded by episodes in which
the male and female touched face-to-face. Touching face-to-face occurred also during
male-male interactions of each species.
Various aspects of touching with faces differed among the species. While touching
females with their faces, male At gregalis generally made stroking movements with their
110
R.R. Jackson
Fig-2. — Mallos trivittatus male taps silk with legs I just before touching female (on
right, facing away from male, abdomen only in view).
Fig-3. — Mallos trivittatus male (Ieft) and female (right). Interplay of legs.
Fig. 4. — Mallos trivittatus male (left) and female (right). Touch with faces. Male’s palps
extend forward and stroke female. Male’s chelicerae open, in preparation to bite
female’s‘phelicerae. ‘ il !
Fig. 5. – Mallos trivittatus male (right) begins to move underl female (left) just before
copulation. Female elevates céphalothorax. Sagittal plants of the two spiders are
angled 45° to 90° to each other.
Osurtship and Dictynidae social organization
112
R.R. Jackson
palps touching the females. Virtually any part of the female’s céphalothorax and abdo-
men might be touched, although the most common location was the female’s face and,
just before or after copulation, her ventral abdomen.
Males of At trivittatus moved their palps during touching also, but in this species
the movements were of greater amplitude than in M. gregalis. When touching the female’s
face, the male tended to wedge his moving palps between those of the female, stroking
her chelicerae with the dorsal surface of his palpal tarsi. The palps were sometimes
extended forward somewhat (fig. 4). Males sometimes extended their palps as they appro-
ached females before touching. During the approach the male’s palps sometimes rotated.
(Each palp rotated in the opposite direction from the other.) When touching each other’s
faces, both spiders tended to have their fangs partially opened (i.e., lowered from the
basal segments of the chelicerae). Males were seen with their faces touching the faces
and the ventral bodies of females, and the latter occurred just before copulation only.
When IX calcarata males touched females with their faces, their palps were held at
the side of the céphalothorax. The fangs were held open somewhat. Almost any part of
the female’s abdomen or céphalothorax might be touched, but by far the most common
was for the male to touch the female’s chelicerae with his own. Both spiders tended to
have fangs open somewhat. The male’s chelicerae were extended forward by tilting his
céphalothorax dorsally and posteriorly with respect to the abdomen. Sometimes the male
simply touched the female briefly with his chelicerae then backed away before repeating.
Other times, he stroked her face with palp movements of relatively small amplitude and
slow frequency, as in AT. gregalis. The distal ends of the male’s chelicerae touched the
anterior surface of the female’s. Sometimes the male also placed his anterior cheliceral
surface under the distal ends of the female’s chelicerae, such that the female’s fangs rested
on his chelicerae. From this position the male moved his palps up and down, stroking
the female’s chelicerae.
13. Bite.
The fangs of At trivittatus (fig. 4) and D. calcarata males sometimes moved some-
what while they were held against the female’s chelicerae. More conspicuous forms of
biting, associated with lunging, will be described for D. calcarata females and At tri-
vittatus males. Often during touching with face, a male X). calcarata would position his
chelicerae around a palp or chelicera of a female, and the chelicerae of the male some-
times closed against the female’s for approximately 1 sec.
14. Lunge.
Sometimes males of At trivittatus stationed themselves immediately in front of
facing females and Intermittently lunged forward with their chelicerae open and palps
spread apart, touching the female’s face momentarily at the most forward part of the
lunge. Between each successive lunge, the male paused for a sec. or longer. At first the
male might not be sufficiently close for lunges to bring his face into contact with the
female’s. Later his face might be continually in contact with the female’s, with the for-
ward part of the lunge serving to briefly push against the female’s face.
Females of D. calcarata sometimes lunged, with chelicerae open, toward facing
males. On the forward part of the lunge, a palp or chelicera of the male sometimes was
positioned between the female’s chelicerae; and the female sometimes closed her cheli-
cerae against the male for approximately 1 sec. or less before backing away. This also
113
happened once in M trivittatus, but the other way around, with the male lunging at the
female and briefly biting a palp or chelicera.
15. Push.
During pushing, two spiders touched face-to-face; and one or both walked or attem-
pted to walk forward, causing the other spider to move backwards. Usually it was the
female that pushed the male backwards during courtship. Pushing was seen during male-
male and female-female interactions in each species, also.
16. Ram.
During male-male interactions D. calcar at a were once seen repeatedly running to-
wards each other, over a distance of a few mm and bringing their open chelicerae into
contact briefly before backing away. This behavior had a rather violent appearance.
H. Organization of behavior during interactions in the laboratory
Courtship behavior can be divided into two phases depending on whether the two
spiders were in physical contact with each other. Interactions normally began with non-
contact behavior, such as the male plucking on the web and the female charging or
lunging toward the male. The contact behavior that followed included stroking with the
palps and legs, biting, interplay of legs, and similar behavior. Copulation sometimes
followed contact behavior, or there could be repeated episodes of non-contact behavior.
Also, episodes of copulation might be interspersed with periods of additional contact or
non-contact courtship; and there were sometimes periods of courtship after copulation,
not followed by further copulation. Frequently the females were inside or near their nests
during courtship and copulation. During courtship, either the male, female, or both
sometimes entered and departed the nest a number of times.
As in most spiders, vision seems to be of little or no significance during courtship
in dictynids. A reasonable hypothesis is that communication is primarily by means of
vibratory signals transirutted through the silk during non-contact behavior and by tactile
and chemotactic signals during contact behavior. The possibility of olfactory communi-
cation should be considered also.
Male-male and female-female interactions had considerable similarity to courtship.
Both contact and non-contact behavior occurred, frequently in alternation. Non-contact
behavior included, among other things, plucking on the web and charging toward the
other spider. All three types of interactions shared some elements of behavior, but a
greater repertoire was generally present during courtship. During all types of intraspedfic
interactions, there were frequently interspersed periods during which one or both parti-
cipants were inactive or walking about in the web. Sometimes walking was associated
with frequent pauses and changes of direction, taking an appearance somewhat different
from locomotion that occurred in the absence of a conspecific individual. Durations of
all types of interactions tended to be variable (table 1). However, all intrasexual inter-
actions lasted less than 20 milL, suggesting that these tend to be shorter than courtship,
but more extensive observation is needed.
R.R. Jackson
Species Type of interaction Observation Duration End
i 1 min mate
2 3 min mate
3 4 min mate
4 10 min mate
Male-female 5 12 min mate
6 13 min mate
7 S min interrupt
8 8 min interrupt
19 min interrupt
10 20 min interrupt
11 4 min desist
Mallos trivittatus 12 11 min desist
1 1 min desist
Male-male 2 1 min desist
3 9 min
1 2 min desist
Female-female 2 14 min desist
3 18 min desist
1 3 hr 51 min mate
. 2 8 hr 10 min mate
3 27 min cannibalism
4 5 min desist
Male-female 5 7 min desist
6 10 min desist
12 min desist
8 20 min desist
Dictynacalcarata 9 2 hr 51 min desist
1 5 min desist
* ■ ; Male-male 2 8 min desist
3 15 min desist
1 3 min desist
Female-female 2 6 min desist
3 11 min desist
Table 1. — Duration of interactions. Recorded until beginning of copulation, until canni-
balism occurred, or until the 2 spiders stationed themselves at opposite ends of the
cage and did not interact further for at least 20 min (desist). Interrupt: when the pair
adopted the copulatory position, they were manually separated with a brush before
copulation actually began.
115
III. Mating in the laboratory
I. Posture.
In each case, immediately before copulation the spiders were face-to-face and tou-
ching. Next the female’s body became elevated and the male moved underneath her
céphalothorax and engaged his palpal organ (fig. 5). The postures adopted by the species
observed in this study and those described by other workers for other dictynids (see
table 2 for reference) correspond to GERHARDT and KAESTNER’s (1937) position I,
in which the male moves under the female from her anterior end and his dorsal body
surface may parallel the female’s ventral, or he may make an angle with her body. In the
dictynids in this study, the angle varied within a range from 45° to 90° for a single species
and even during a single copulation. This was surprising since GERHARDT and KAEST-
NER noted that the copulatory angle adopted by a given species tends to be constant.
* Species Observation Duration
Mallos gregaUs i 100 min.a
2 1 : >• 21 min.b
3 11 min.b
Mallos trivittatus f§ 1 8 hr.c
2 60 min.
3 47 min.
4 39 min.
5 28 min.
6 22 min.
Dictyna calcamta 1 78 min.
2 43 min.
Dictyna civica BILLAUDELLE (1957) 14 min.
Dictyna la tens Locket (1926) 15 min.
Dictyna uncinata Gerhardt (1924) 30 to 60 min.
Dictyna volupis Montgomery (1903) 1 to 2 hr.
Ixeuticus longinuus Gregg (1961) 1 to 2 hr.
a Performing non-contact courtship when first observed. Possibly copulation had
preceded observation period.
b Copulation in progress when observation began.
c Estimated.
Table 2. — Copulation durations in laboratory, recorded as time during which male palps
were applied to female epigynium (intervals of other behavior between periods
of palp application deleted). Numbered observations were from the present
study.
116
R.R. Jackson
Also, the males of these species rotated their bodies during copulation such that the
sagittal plane of the male and that of the female made an angle with each other ranging
from 0° to 90° (fig. 5). I am not aware of previous reports of this sort of rotation during
copulation in spiders. The male’s legs I and II tended to be touching the female’s body
during copulation, and their exact positions were variable, but in general they wrapped
around the female’s céphalothorax and abdomen. Frequently there were periods during
which the male rhythmically pulled on the female’s body with these legs while the palpal
hematodocha pulsated.
2. Duration.
Durations of copulations seen in this study, plus those reported in other studies, are
given in table 2. Since there tends to be considerable intraspecific variability, more exten-
sive data are needed in order to clarify how durations vary interspedfically. The longest
copulation (8 hr; At trivittatus) was an estimate, since I was away from the laboratory
inadvertently on three occasions during this time (45 min, 35 min, 15 min). During the
time of observation, there were interspersed short periods of courtship and inactivity,
especially in the late afternoon, totaling approximately 30 min. BlLLAUDELLE (1957)
reported observing more than 50 copulations of Dictyna dvica9 each of which lasted
14 min. This remarkable consistency contrasts with the great variability observed for the
species in this study.
3. Pattern of palp application.
The dictynids apply one palp at a time. The pattern in which the two palps are used
seems to be variable. For example, in the case of observation n° 1 for D. calcarata
(table 2), one palp was applied for 75 min, then the male walked away from the inactive
female, and resumed courtship, with frequent periods of inactivity. Almost 1 hr later,
the other palp was engaged for 3 min. After this the female became active, and the pair
separated for the final time. During observation n° 1 for M gregdis there were inter-
spersed episodes of contact courtship between palp engagements, and the same palp was
engaged twice in succession before switching to the opposite ride. A single palp engage-
ment occurred during the other two observations for M. gregatis. With one exception,
during each copulation involving M. trivittatus and D. calcarata, each palp was applied
only once, and there were periods of courtship between each palp application. The excep-
tion was the 8 hr copulation of a pair of At trivittatus that was not observed in its
entirety. Three applications of the left and four of the right palp were observed, and
once the right palp was applied twice in succession* |
4. Receptivity.
Males of each species studied in the laboratory readily mated with more than one
female. Sometimes previously mated females of both D. calcarata and At trivittatus re-
mated with a different male on another day, after having become gravid with eggs. In
one instance, I separated a pair of At trivittatus (female had mated previouriy with
another male) with a brush just as they began to copulate* The female subsequently
oviposited fertile eggs, demonstrating that females of this species will mate with a second
male even while carrying fertile sperm from a previous copulation. Females of D. calcarata
and At trivittatus will also mate after opposition. Information is not presently available
concerning At gregalis females; and the relation between insemination, opposition, and
receptiPty is generally poorly understood for spiders.
Courtship and Dictynidae social organization
117
IV. Observations from the natural habitats of the spiders
Because observations of intraspecific interactions in the field are comparatively rare
for spiders, those recorded in this study are reported individually in table 3.
It has been proposed that males of communal, territorial species are more nomadic
and females are more sedentary, with males walking about within the web complex or the
general environment rearching for females (JACKSON, 1978b). The same hypothesis
would seem to apply to solitary species. Some observations relevant to this hypothesis
will be mentioned here. In a large web complex of AT. trivittatus in Arizona, estimated to
contain 10,400 spiders, 11 males, compared to 6 females and 4 immatures, were seen
walking. The estimated ratios of each sex/size class in the web complex were 9.3% males,
26.7% females, and 64.0% immatures (JACKSON and SMITH, 1978). On two separate
occasions, males of the solitary species D. tridentata Bishop and Rudeman were seen
walking on an outdoor table, not in the immediate vicinity of webs. Another six males
were found inside webs. In contrast, all 25 females and immatures of .this species that
Vere found were located inside webs.
V. Spinning behavior during courtship
. • . < .
BRISTOWE (1958) reported that males of Dictyna L. construct a
special “chamber” or “mating canopy”, presumably with similarities to the nest of the
species in this study, inside which the pair copulate and later reside for a period of a
month or more. The spinning behavior of D. tridentata males (table 3) suggests the
possibility of something similar in this species. Also, once a male M, gregalis was seen
spinning at a nest, with a female standing approximately 1 cm away; and once a male
M. trivittatus spun briefly during courtship in the laboratory. In each case, the male spun
rapidly, with frequent changes in direction, and sperm webs were not constructed. Since
spinning is an integral part of courtship in some species of Thomisidae, Salticidae, Aranei-
dae, and other groups of spiders (BRISTOWE, 1958; GRISWOLD, 1977; JACKSON,
1977b), the possibility that spinning in dictynids has a communicatory function should
be investigated.
VI. The role of sexual dimorphism in communication
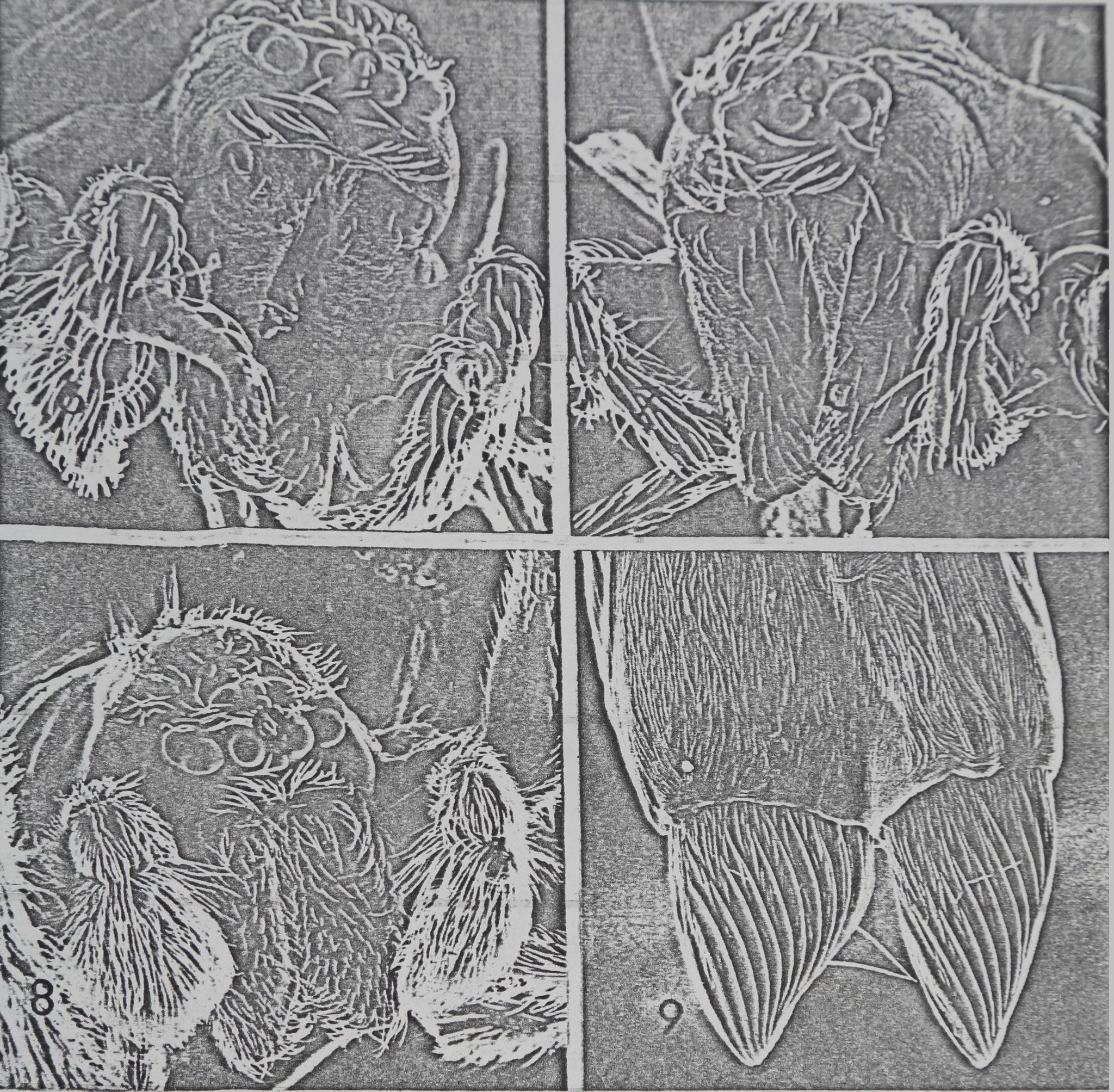
Pronounced sexual dimorphism tends to occur in the shape and size of chelicerae
of adults in the genus Dictyna (CHAMBERLIN and GERTSCH, 1958), those of males
being larger and more curved (compare fig. 6 and 7). Females of Dictyna walckenaeri
Roewer use their chelicerae to grip those of the males (BERLAND, 1916; quoted vice
versa by BRISTOWE, 1958). The chelicerae ofD. calcarata are bow-shaped, and those of
males are larger and more curved than those of females (fig. 6 and 7). In contrast, the
chelicerae of Af. gregalis and M. trivittatus are not bow-shaped (fig. 8). Males of AL
trivittatus and both males and females of D. calcarata sometimes placed their chelicerae
around a palp or chelicera of another spider during courtship. None were seen to place
their chelicerae around both chelicerae of their partners simultaneously, as in D. walo
kenaeri Further studies should consider the possibility that differences in cheliceral shape
00
1 – Species Type of interaction Observation Elements of behavior Description
Male-female 1 1 ill Twitch abdomen (M), pluck and and tap silk (M), interplay of legs (M & F), charge (F), chase (F). M repeatedly returned and courted after F chased.
Male-female 2 Same as 1 Same as 1
Male-female 3 Same as 1 Same as 1
Male-immature I 4 Twitch abdomen (M), pluck and tap silk (M), interplay of legs (M & I), chase (I).1 I (6mm), initially inside nest, departed nest to chase M.
Male-male 5 Twitch abdomen (n.2), pluck and tap silk (n.2), interplay of legs (n.l & n.2), chase (ml)1 M n.l inside nest initially; M n.2 outside, n.l departed nest to chase n.2.
Mallos trivittatus Immature-immature 6 Chase (n.l)1, : I n.l (4mm), I n.2 (3mm)
Female-female 1 | 7 | | Interplay of legs (n.l & n.2), chase (n.l)1 Both on “extension line” (JACKSON, 1978a), F n.l closer to nest.
Female-female • 8 Interplay of legs (n.l & n.2), chase (n.l)1 Large diptera (Tipulidae) lands on web. n.l departs nest and begins to feed. n2 arrives from neighboring web unit 10 sec later
Female-immature 9 Chase (F)1 Large diptera lands on web. F departs nest and begins to feed, i I arrives from interstitial area.
R.R. Jackson
Dictyna calcarata I – Male-male 1 Twitch abdomen (n.l & n.2), push (n.l & n.2), chase (n.l)1.
Male on empty web unit 2 Twitch abdomen, pluck and tap silk Enters nest, walks inside briefly, then departs web unit.
Mallos niveus Male-female if copulate Male-female angle: 90°
Dictyna tridenta Male-female i Twitch abdomen (M), touch with face (M & F), push (F), chase (F) M spins in and near nest. F feeds on Diptera, 8°” away. F departs prey and goes to M. After chase, F returns to prey and M returns to spinning near nest.
Male-female 2 Same as 1 Same as 1, except’no prey presen present. F mostly inactive, a few cm from M.
1 Chased Individual departed web unit and did not return. Chasing individual either did not depart the web unit or departed and then returned.
Table 3. Interactions obseived in nature. Elements of behavior described in text. Body lengths of immatures given in
parenthesis. M: adult male, F: adult female, I: immatures.
a
Courtship and Dictynidae social organization
Fïg. 6. — Face of Dictyna calcarata male (scanning électron micrograph), c: Bow-shaped
chelicerae. o: Ocular region of céphalothorax, h: Hematodocha. s: Palpal spur
(enlarged in fig. 9). Distance between inner of two chelicerae at widest point of
bow: 100 vut ^
Fig- 7. — Face of Dictyna calcarata female (S.E.M.). o: Ocular region, c: Chelicerae (less
bow-shaped than those of male, fig. 6). Same scale as fig. 6.
Fig. 8. — Face of Mallos trivittatus male (S.E.M.). o: Ocular region, c: Chelicerae (not
bow-shaped, as on Dictyna calcarata, fig. 6 and 7). Distance between the inner edges
of two antero-medial eyes: 100 u.
Fig. 9. — Tip of palpal spur of Dictyna calcarata male (S.E.M.). Diameter of spur at vppcr
end of photograph: 50 u.
Courtship and dictynidae social organization
121
constitute signals during touching with faces and biting during the courtship of dictynids.
Another striking case of sexual dimorphism in D, calcarata is the spur on the palpal
tibia of the males (fig. 7 and 9). Although it seems probable that this structure is involved
as a signal, there is little evidence as to function at this point. Usually, when spiders
touched faces, the male’s palps were held at the side of the chelicerae in a position such
that the spurs pointed dorsally and did not contact the other spider. However, when the
male: positioned his chelicerae under those of the female and stroked her with his palps,
the spurs rubbed against the female’s chelicerae. Also the spurs contacted the female’s
abdomen when the male touched the female with his face just before copulation. The
relatively few observations made on this species should be emphasized; and we should
be prepared for the possibility that the spurs are employed, on a relatively infrequent
basis, in a more distinctive fashion. For example, in the salticid spider Phidippus johnsom
Peckham and Peckham, some of the more distinctive elements of courtship behavior
occurred in only a small fraction of the observed interactions (JACKSON, 1977b).
_ ‘ VII. Aggression
1. General comments.
Since the behavior of males during male-female interactions was associated with
mating or attempts to mate, this was referred to as “courtship.” Interactions involving
other combinations of spiders (male-male, etc.) led to one individual departing from the
vicinity of the other, and the behavior involved will be referred to as “aggressive”. This
type of behavior occurred readily when male-male and female-female pairs of Af. trivittatus
and D. calcarata were placed together. In general, spiders of these species will not
tolerate close proximity of other individuals of the same sex and size class (JACKSON,
1978a).
In contrast, aggressive behavior was extremely rare in Af. gregaüs. On two occasions
I observed male-male pairs pushing and chasing (another observation, SJE. SMITH, peiso-
nal communication), but usually males that encountered each other simply walked away.
Behavior of females, such as charging and chasing males, seems to be related to unrecep-
tivity and interference with mating attempts by the male. These motor patterns can be
called “aggressive,” and this type of aggressive behavior was seen in Af. gregaUs, as in the
other two species. In general, individuals of all sex and age classes of Af. gregalis lived in
close proximity/ frequently touching and walking over each other, without apparent
aggressive behavior.
2. Aggressive behavior in the presence of prey.
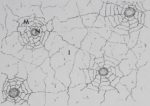
In A£ gregalis, groups of spiders routinely fed together on the same prey. Sometimes
flies became completely covered by feeding spiders (fig. 10). Individuals that arrived after
this point walked around on the feeding spiders, making no attempt to drive away other
individuals to make room for themselves, and eventually simply walked away.
In M. trivittatus, sometimes more than one spider fed on the same prey item.
However, unlike the situation of Af. gregalis, this was the exception instead of the rule in
Af. trivittatus, occurring in only 5 of the 58 observed cases of feeding in the field. In each
case the spiders were relatively widely spaced around the prey. 1. A female and male fed
at opposite ends of a tipulid fly (3 011 apart). 2. A male and an immature (body length:
122
R.R. Jackson
5mra) fed at opposite ends of a Diptera (10X1 ). 3. A male and two immatures (each 2mm)
fed on a tipulid. 4. A female fed with an immature (31,1,11 ) on a tipulid. 5. Another female
fed with an immature (3mm) on a tipulid. In this case, prey capture was witnessed. The
fly landed on the web and became stuck. The female rushed out and began to feed, fol-
lowed soon by an immature from the same web unit. An aggressive interaction followed
in which the female pushed and chased the immature; the immature returned repeatedly;
and the female eventually tolerated the immature at the fly. Other aggressive interactions
involving prey are summarized in table 3.
In each of 13 observations of feeding in nature by other species (M. niveus, AL
dugesi Becker, D. calcarata, D. tridentata, D. compléta Chamberlin and Gertsch, D.
phylax Gertsch and Ivie), only one individual fed on a given prey (JACKSON, 1977c).
However, it seems likely that feeding in small groups occurs occasionally in other com-
munal territorial species, as it does in M. trivittatus, rince joint feeding by a male-female
pair of Z). calcarata was seen in the laboratory. A male-female pair of D. calcarata was
kept together, observed intermittently, and fed houseflies (Musca domestica) for 14 days
after mating. Generally the spiders remained on opposite rides of the cage, although once
they were observed feeding on the same fly. Feeding in pairs may also occur in some
solitary species, in which males share webs with subadult and adult females (JACKSON,
1978c). Although not yet seen in the spedes in this study, BRISTOWE (1958) reported
that male-female pairs of some European spedes will feed together on the same prey.
Fig. 10. — Group of Mallos gregalis feeding on the same fly. f: Adult female, i: Immature.
123
Courtship and Dictynidae social organization
VIII. Spacing tendencies
1. Introduction and methods for recording spacing.
In M. gregaUs, individuals of all sex and age classes were generally seen in close
proximity of other individuals. InM. trivittatus and H calcamta greater distances between
individuals usually occurred. This difference is probably related to differences in aggressive
behavior. In the laboratory, a systematic observation procedure was devised for recording
the differing spacing tendencies of these species.
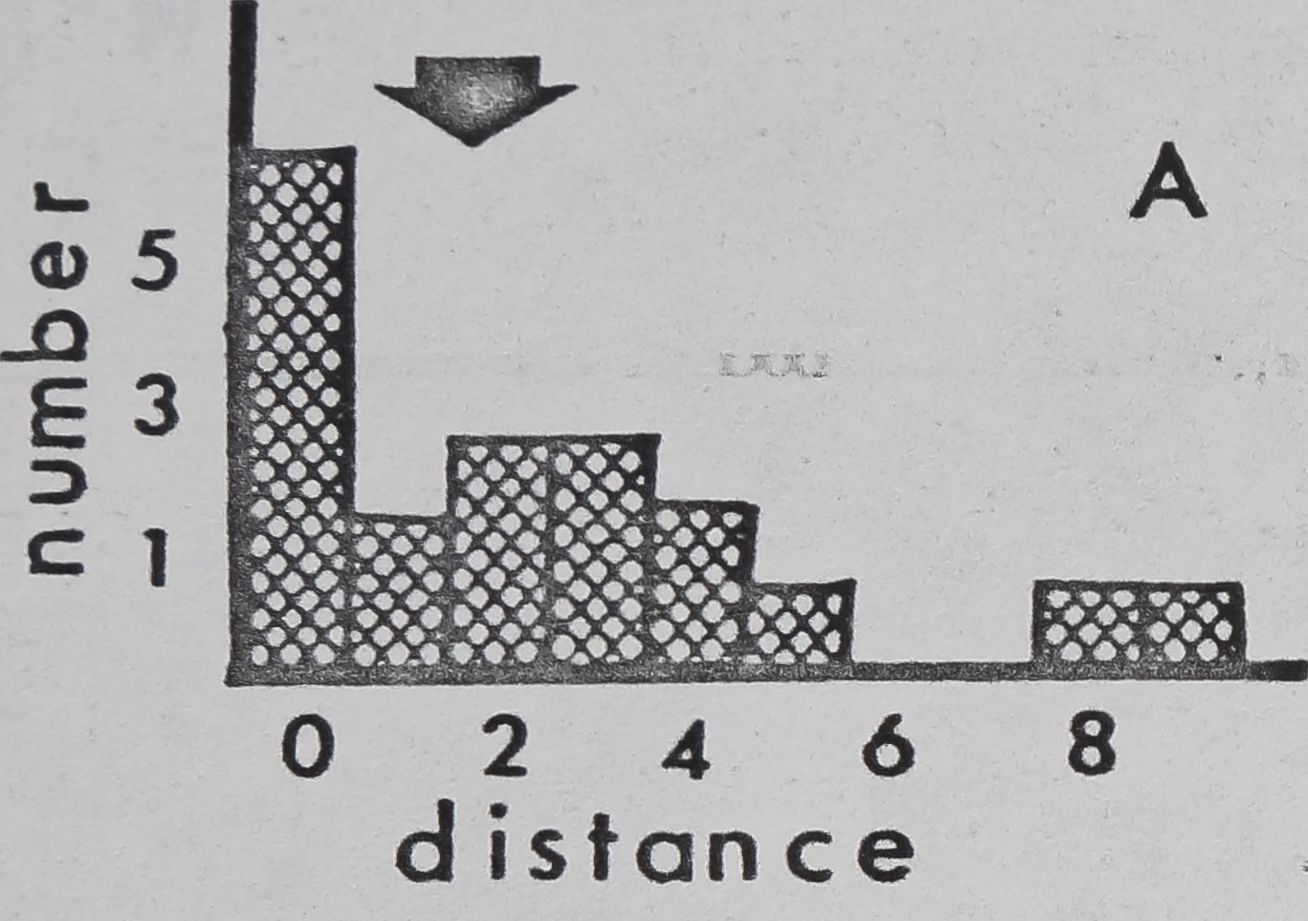
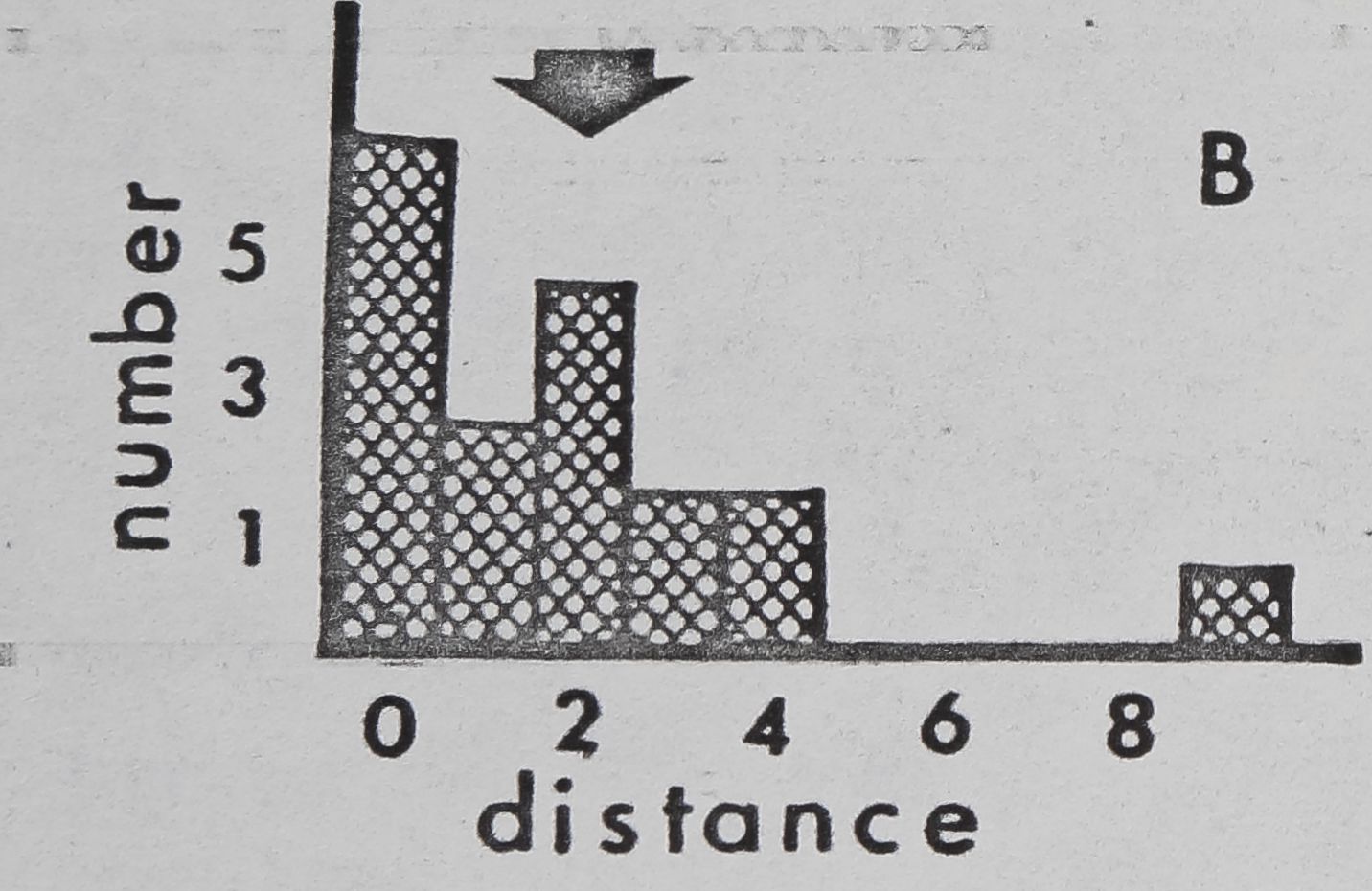
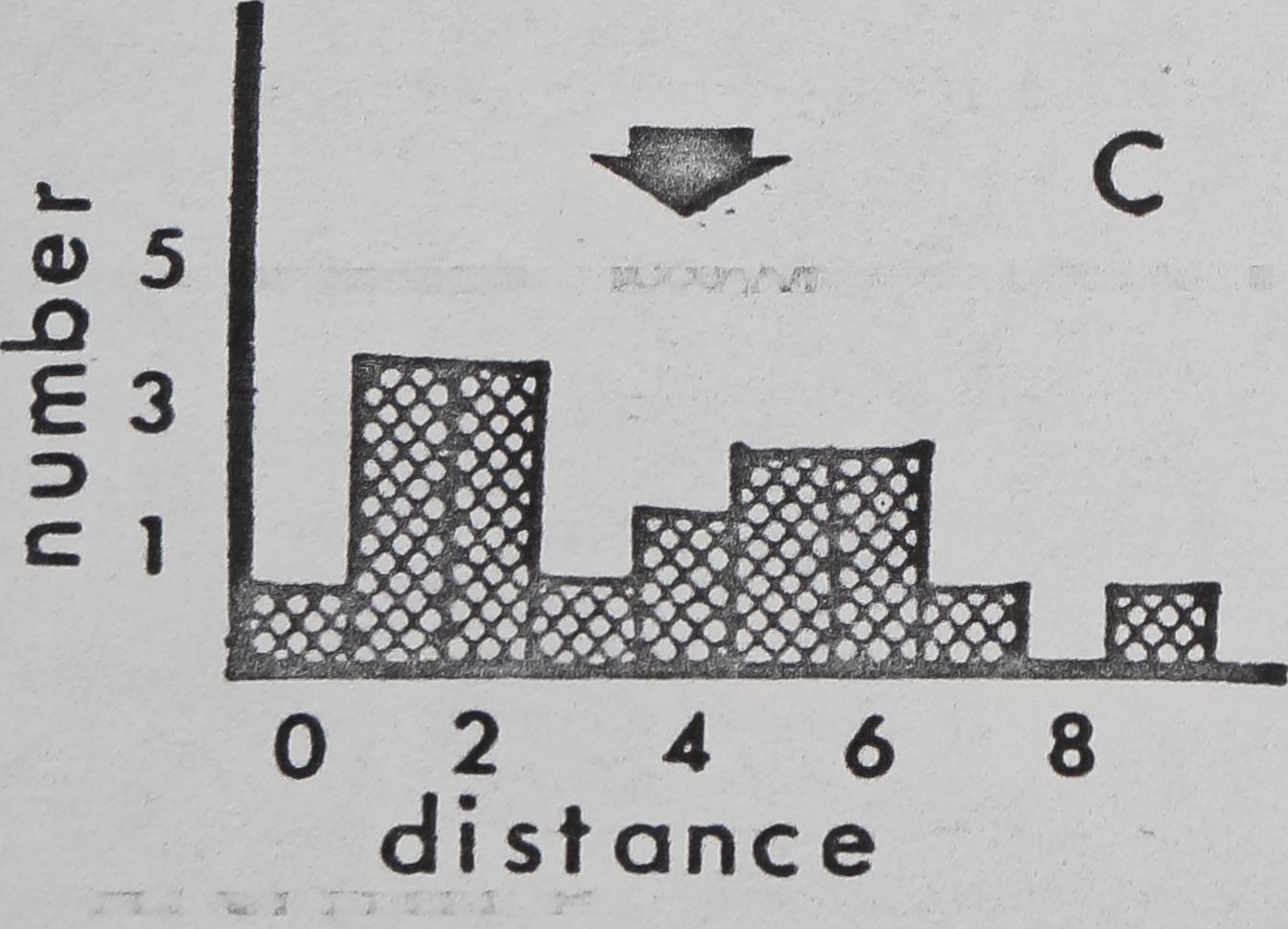
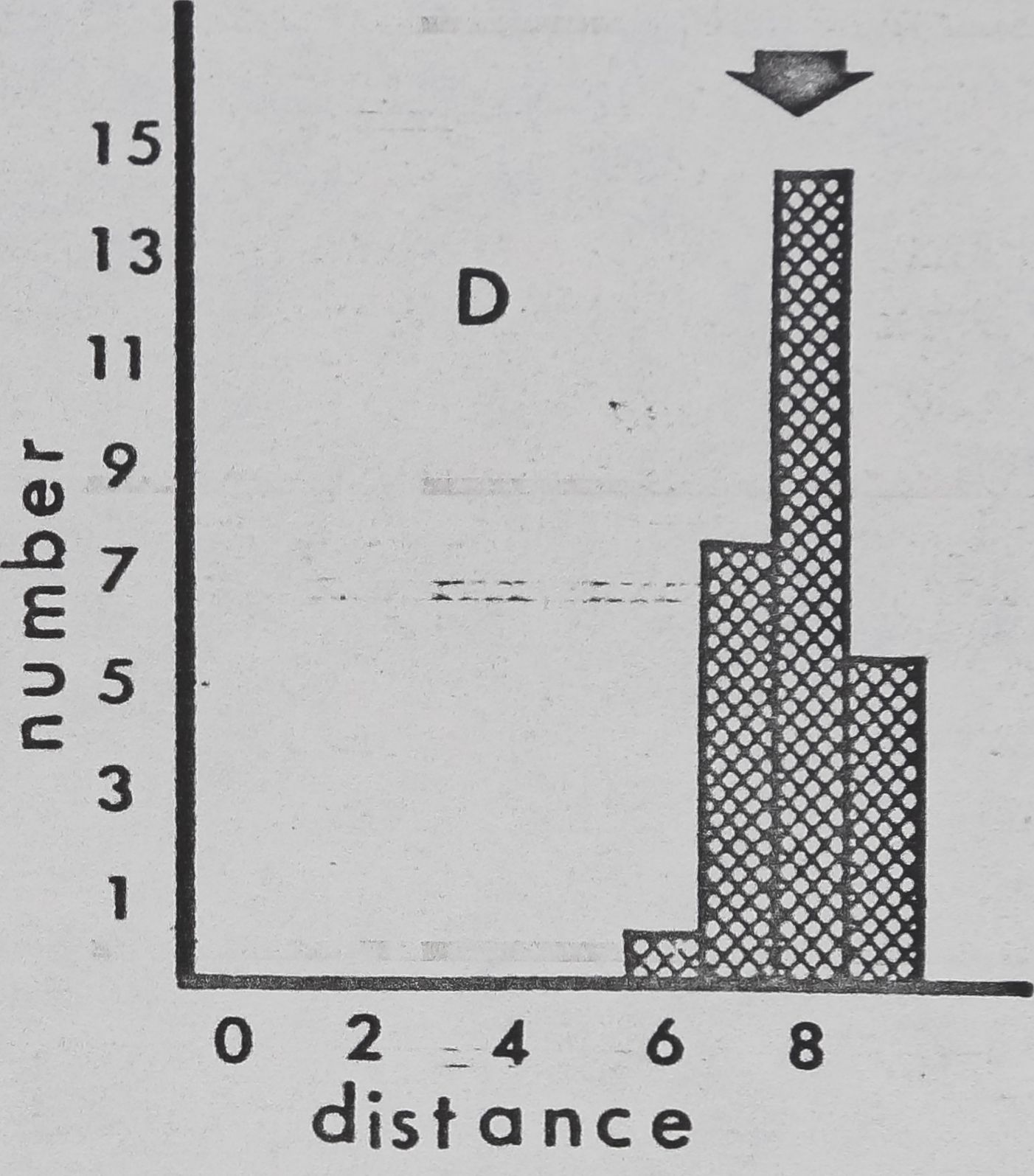
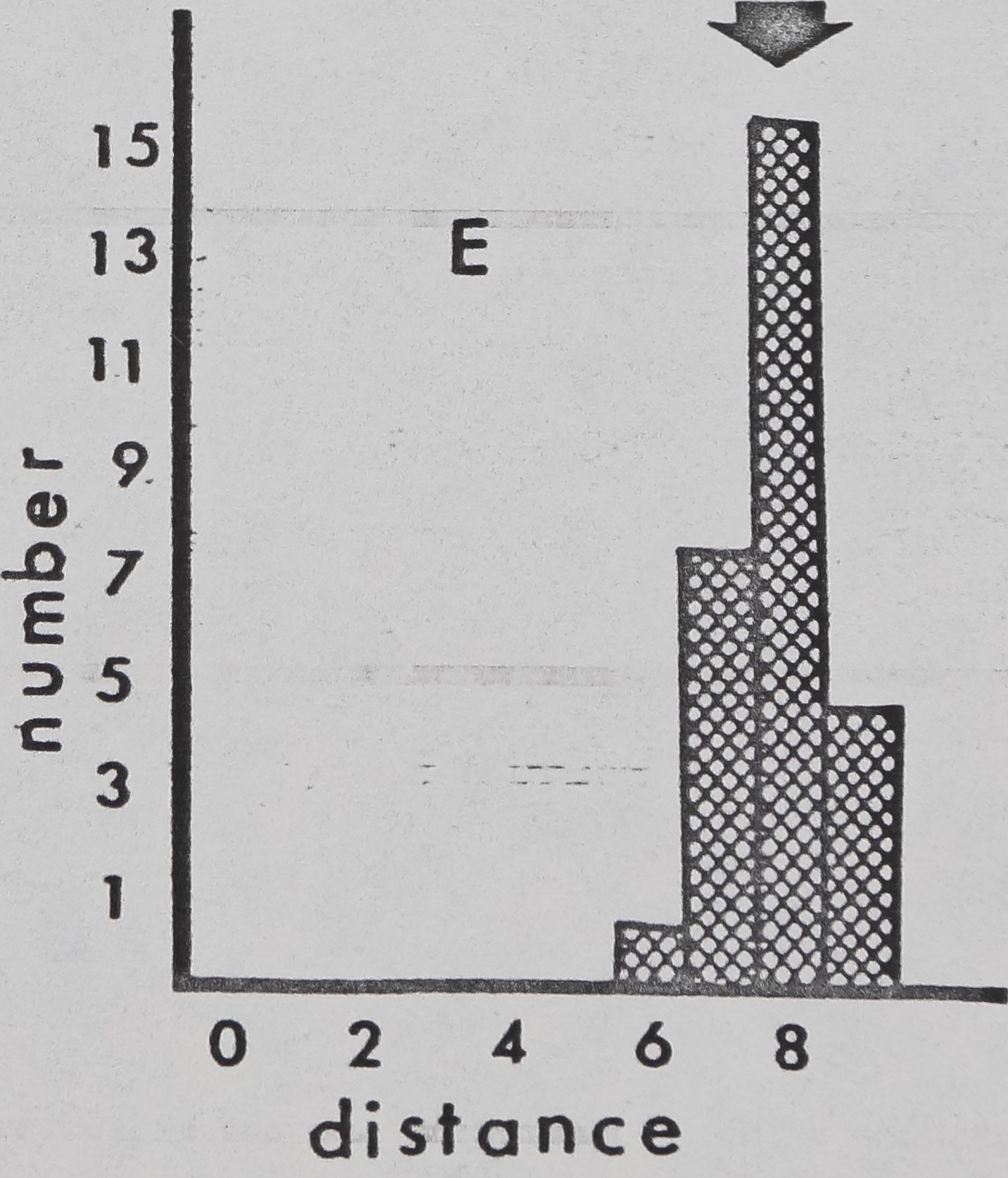
Fig. 11. — Interindividual distances of pairs of spiders in laboratory cages (cage diameter:
9cm). Mean for 4 observations, recorded to nearest cm, was used for each pair. 0:
Members of pair in physical contact. 1: Members of pair 1^ or less apart, but not
touching. 2: Members of pair between 1 and 20,1 apart. Etc. Arrows: means for all
pairs. A: Mallos gregalis, adulte female, 20 pairs. B: Mallos gregalis, immatures, 20
pairs. C: Mallos gregalis, adult males, 20 pairs. D: Mallos trivittatus, pooled data for
each sex/age class very similar. E: Dictyna calcarata, pooled data for 10 pairs each of
adult females, immatures, and adult males. Data for each sex/age class were very
similar.
124
R.R. Jackson
Cages were of the same type used for maintenance (fig. 1), except that there was
no cotton roll or Drosophila culture, and all four holes were plugged with corks. Two
individuals of the same species and age/sex class were placed inside each cage at 1400 hr.
On the following day, a record was made of the distance apart of the two spiders in each
cage at 1100 hr, 1300 hr, 1500 hr, and 1700 hr. (Lights on, 800 hr; off 2000 hr).
2. Results and discussion.
During the day, spiders were relatively inactive. Only three were seen walking at
the time of observation. Variation in the distance apart for a given pair through the day
was small (mean within pair S.D.: 0.72e111).
Pairs of M. gregalis spaced more closely together than At. trivittatus (P(0.001) and
D. calcarata (FCO.Ol ; Mann Whitney; pooled data for all sex/age classes)(fig, 11). This will
be interpreted as a tendency of At. trivittatus and D. calcarata to maintain relatively large
individual distances (HED1GER, 1950) with respect to members of the same sex. AL
gregalis females and immatures (pooled data) space more closely than males (P(0.01;
Mann Whitney). Close spacing by females and immatures in these observations is probably
related to a tendency of these spiders to aggregate and live in high densities on communal
webs. The significance of wider sparing by males of At. gregalis may be related to sexual
competition, since maintenance of an individual distance by a male might reduce inter-
ference by other males during courtship and mating. However, interference of this type
has not been seen thus far in.this species.
In nature, small immatures of At trivittatus and D. calcarata were sometimes situ-
ated Ie111 or closer to larger immatures and adults. Adult male-female pairs sometimes
shared web units. Males of At. gregalis might be expected to space more closely to females
than to other males. Future studies should explore the spacing relations of heterosexual
pairs and groups of immatures of varying sizes with and without adults, j – * – ?
DL Cannibalism ^ t
-‘»• – pjj -* : * -r .-.-4* _ • . –
1. Miscellaneous observations. ; _
Observations in nature and the laboratory indicate that while other dictynid species
tend to be cannibalistic, ML gregalis is almost never cannibalistic.*In the communal webs
in the laboratory,* only two instances of cannibalism involving At. gregalis have been seen,
despite a great many hours spent by several investigators observing these spiders. How
the two cases of cannibalism came about is unknown. One case was a female feeding on a
male, and the“ öfter was a female feeding on an immature. Once a small communal web
in a 10x10x7^. plastic cage, containing approximately 20 individuals of all sex/age
classes, was kept without flies (water provided) for three weeks and watched casually.
Although the spiders frequently touched and walked over each other, there was no
evidence of cannibalism. P.N. WlTT maintained a similar communal web without flies
for. 4 weeks without seeing cannibalism (personnal communication).
In contrast to AL gregalis, both direct and indirect evidence of cannibalism was
relatively common for other dictynid species. 1. During a male-female interaction in the
laboratory, a female D. calcarata killed and ate a male after 27 min of courtship.
Courtship and Dictynidae social organization
125
Immediately beforehand the female had been pushing the male. Then she moved over
the male and inserted her fangs into his carapace. The remaining observations were from
the field. 2. Twice adult females of Af. trivittatus were seen feeding on other adult females
of approximately the same size. 3. Three dead females and two dead immatures were
seen, each in a different web unit of A£ trivittatus. In each case, an adult female occupied
the web unit; the dead spiders had the appearance of eaten prey (dry; collapsed abdo-
mens); and the dead spider was at the periphery of the web. The remaining cases involve
solitary species. 4. A dead male was found in a web with a female of Dictyna sp.
(Querecho Plains, JACKSON, 1978a) and her eggs. 5. A dead male of Af. niveus was
found in a web with another male. 6. A dead female of Af. niveus was found in a web
with a male. Each of these dead spiders had the appearance of eaten prey.
3. Methods for recording cannibalism.
An observation procedure was devised for comparing the cannibalistic tendences
of different species. Cages were of the same design as those used for observations of
spacing behavior. An adult female and two immatures (1 to 3mm in body length) were
placed into each cage. No flies were provided. Each cage had spiders of one species only.
(Previous to these observations, each spider had been kept in an individual cage without
food for 2 days.) For two days afterwards each cage was checked once in the early
morning, once in the early afternoon, and once in the early evening (using a flashlight).
Sixteen cages were set up for AC gregqlis (communal, non-territorial species), 16 forAf.
trivittatus (communal, territorial), and 4 for AC niveus (solitary).
3. Results.
Two females of AC trivittatus were seen feeding on immatures. In another 7 cages
one or two (one cage) immatures of AC trivittatus were found dead. Cannibalism seems
likely in these cases since the carcasses had the appearance of eaten prey. In contrast, no
deaths occurred for AC gregalis during the observation period. The frequencies of cages
in which deaths of spiders occurred were significantly greater for AC trivittatus (X2=9.899,
1X0.005). Also, one of the cages of AC niveus had a dead immature, apparently having
been preyed upon.
Discussion
I. Interspecific differences in courtship
The descriptions provided in this study should be viewed as a preliminary report,
since there was considerable intraspecific variability and only a few interactions were
observed for each species. In some animals there are elements of communicatory behavior
that are used only infrequently, and a substantially more complete view of the species’
communicatory behavior can be obtained by increasing observation time (ALTMANN,
1968; DANE and van der KLOOT, 1965; JACKSON, 1977b). However, with these limi-
tations in mind, it would be valuable to compare the courtship of the three species
observed in the laboratory in this study.
Abdomen twitching, and touching with face were of different form in each species.
Interspecific differences in plucking and tapping silk, interplay of legs, pushing, and
biting, although not clearly discerned, deserve further study. Jerk and quiver were obser-
ved in A/, trivittatus only; rock in D. calcarata only; and pluck walk in Af. gregalis only.
R.R. Jackson
II. Elements of behavior in other species
Apparently, the elements of behavior observed for the species in this study are
similar to those that occur during intraspecific interactions in other species of Dictynidae
and spedes in the closely related family Amaurobiidae (BERLAND, 1916; BILL AU-
DELLE, 1957; BRISTOWE, 1958; GERHARDT, 1924; Gregg, 1961;Locket, 1926;
McKEOWN, 1936; Montgomery, 1903). However, since descriptions of spider court-
ship are generally lacking in detail, it is difficult to use the literature for comparative
purposes.
III. The non-cannibalistic nature of MalJos gregalis
Mallos gregalis are non-cannibalistic despite seemingly ample opportunity. For exa-
mple, when individuals arrived at a fly that was already covered by feeding spiders (fig.
10), they never inserted their fangs into al eg of another individual and treated a conspedfic
as prey. This would seem simple enough to do; and the cannibalistic individual would
seem to benefit by gaining a meal and eliminating a competitor, which are important
selection factors in some organisms (FOX, 1975). These observations raise the question
of possible disadvantages for cannibalistic individuals. Perhaps cannibalism is selected
against by kin selection (HAMILTON, 1964) orinterdemic selection (WRIGHT, 1960).
However, at this point we know almost nothing about the relatedness of individuals in
natural populations or the rates of local population extinction for M. gregalis. More
importantly, it is not clear that any of the characteristics, including the lack of canniba-
lism of these spiders require explanations outside the realm of individual selection.
Possibly there is a risk involvèd in making a predatory attack on another spider,
since the attacked individual might injure or kill the attacking individual in the course
of defending itself. Also the web is quite important in subduing prey (JACKSON, 1978d)i
Since M. gregalis do not become stuck in their own webs, another conspedfic individual
might constitute an especially difficult prey compared to a fly caught in the web. The
energetic costs entailed in waiting for another fly caught in the web may be less than
the costs, both in energy and risks, entailed in subduing another conspedfic not caught
in the web. This would be espedally true if these spiders have substantial immunity to
the venom of conspedfics, because in this case the bite would not quickly immobilize
the victim. Since it seems unlikely that the attacked individual would remain stationary
sufficiently long for the attaking spider to inject a great volume of venom, the venom
would need to be rather effective. Physical restraint seems unlikely except for an adult
attacking a very small immature, since immobilization wrapping (see ROBINSON, 1975)
of prey does not occur in the Dictynidae, and these spiders do not seem to use their
legs in holding prey (see ROVNER, 1978).
Although dead and not yet dried out individuals were seen at times in communal
webs, feeding on these was never seen even though they were touched and walked over.
Spiders will feed on dead, not yet dry, flies that they locate in the web. These observa-
tions raise the possibility that these spiders cany a toxin in their tissues that renders
them noxious to other individuals. However, an immature Fhidippus audax Hentz (Ara-
neae, Salticidae) in the laboratory readily fed on M. gregalis taken from their web.
Courtship and Dictynidac social organization
127
providing no evidence that ML gregalis is protected by a toxin at least in the case of this
predator. It is important to bear in mind the distinction between a toxin that might
constitute a selection pressure against cannibalistic individuals and a chemical that serves
solely as a signal (pheromone).
IV. The non-aggressive nature of Mallos gregalis
In general, speculations concerning the absence of cannibalism do not apply equally
well to the absence of aggressive behavior.
One might predict that an individual who forces another away from a fly, at which
there is no space to feed, would be at a selective advantage. However, conceivably the
energetic costs and/or the risks of injury, if the opponent defends itself or escalates the
intensity of the interaction (MAYNARD SMITH and PRICE, 1973), are greater than
the costs involved in waiting for another fly.
In the laboratory, food was probably not limiting since the spiders were fed gene-
rously at frequent intervals; and this may be rather similar to the conditions in the
spiders* natural habitats in Mexico during the rainy season. DlGUET (1915) and
BURGESS (1976) made their observations in Mexico during the rainy season. What
happens when the dry season arrives and food becomes less plentiful is unclear, since
the few observations that have been carried out on this species in nature were mostly
during the rainy season. The cost/benefit relations of spiders with respect to cannibalism
and aggression would be expected to change with changes in the availability of prey.
DigUET’s (1915) comments suggest that, at the end of the rainy season, some individuals
disperse from the web and others remain at the web in a dormant state. We do not know
whether aggressive and cannibalistic behavior occur with the arrival of the dry season,
but there was no evidence of this in the webs kept in the laboratory without food for
approximately a month. Since spiders are known to be adapted to endurance for exten-
ded periods without food (see ANDERSON, 1974; WITT, 1963), observations over a
longer period of food deprivation would be valuable.
V. The function of courtship
If cannibalism reduction were the only function of courtship in dictynid spiders,
then we would predict the absence of courtship in the non-cannibalistic species A£ gregalis
(see BURGESS, 1976). However,M. gregalis has distinct male courtship. If cannibalism
reduction were a major but not exclusive function of male courtship, then we would
predict less complex courtship in ML gregalis. A procedure of measuring complexity has
not been devised for these spiders, although one approach might be to measure informa-
tion content (SHANNON and WEAVER, 1949) with a larger number of observations.
However, on the basis of the observations made in this study, differences among species
in complexity are not evident. In each species there was repeated performance, in varied
order, or a number of different signals.
If cannibalism reduction were ä major function, then we would predict male court-
ship to have a “cautious” character in cannibalistic species, and we would predict M
gregalis courtship to be less cautious. The question of how to measure “caution” has
not been dealt with, but various aspects of male courtship give an impression of caution
128
R.R. Jackson
in each species. For example, males frequently approached then withdrew from females,
paused when the females began to walk, and moved their forelegs slowly and gently
during interplay of legs. However, interspecific differences in caution were not evident.
GEIST (1971) pointed out that there is an alternative explanation for caution in male
courtship for species in which males are not capable of physically restraining the female.
Assume that the female will not mate until she has monitored some variable duration of
male courtship. A male that attempts to mate, to approach closely, or to progress to a
more advanced stage of courtship (contact phase, e.g., in dictynids) may force the female
into making a **decision” as to whether she will permit copulation, proximity, etc. before
it is optimal for her. Under these circumstances females may be very prone to simply
decamp, ending the courtship interaction. A male that courts cautiously, hesitating when
the female fails to respond, begins to decamp, or responds aggressively may be less likely
to precipitate a premature departure by the female.
Another consideration is that cannibalism reduction lacks generality as a function
of courtship in the animal kingdom, since highly complex courtship occurs in many
groups in which cannibalism by the female would not seem to be a likely selection pres-
sure on the males (e.g.. Drosophila, SPIETH, 1974; grasshoppers, LOHER and CHAN-
DRASHEKARAN, 1972; the niff, RHlJN, 1973). This should caution us against under-
estimating the importance of functions other than cannibalism reduction for spider court-
ship. Certainly courtship might have multiple functions that differ in different groups of
animals, and perhaps cannibalism reduction is an important function of courtship in some
spiders. However, each case for which this is proposed should be critically investigated,
and the primacy of this function for spiders in general is questionable.
1 What then are alternative functions that might be considered for animal courtship?
No attempt will be made to review the extensive literature on this subject (see BASTOCK,
1967; MANNING, 1965; Morris, 1956). Instead, a few specific doting remarks will
be made.
To say that the function of male courtship is to arouse the female or to bring her
into readiness to mate does not completely answer the question, but shifts its focus: what
is the function of a system in which females require male courtship before they are
prepared to mate, or what is the function of female “coyness?” (RICHARDS, 1927;
Manning, 1966).
Two proposed functions have received particular emphasis. PECKHAM andPECK-
HAM (1889, 1890) argued for sexual selection by female choice in spiders. No data are
available for the dictynids relevant to this hypothesis. Reproductive isolation has been
emphasized as a function of animal courttitipby DOBZHANSKY (1970), MAYR (1963),
and others. Reproductive isolation would seem to present a potential problem for the
dictynids in this study. Sometimes more than one species of the same genus were found
in close proximity within the same habitat. For example, sometimes ML niveus were
found on stems of the same trees on which there were M. trivittatus in web complexes
around the trunk, as close as lm away. Also, the mating seasons of these two spedes
overlapped. However, merely to point out interspedfic differences in courtship behavior
does not constitute adequate support of the hypothesis. Courtship between individuals
of related spedes and female responses to experimentally manipulated signals (simula-
tions of male vibratory signals using electronic transducers, alteration of male cheliceral
shape, etc.) should be investigated. However, the most valuable approach would be com-
parative, looking at courtship in populations of the same spedes in which there are
129
differing degrees of sympatry with related species. From the reproductive isolation hypo-
thesis for the function of courtship, we would predict differences in courtship. The
sexual selection hypothesis does not lead to this prediction.
A cknowledgemen ts
For valuable discussions, I would like to thank P.N. WITT, M.C. VICK, S.E. SMITH,
and J.W. BURGESS. Special thanks go to W J. GERTSCH for his assistance in the identi-
fication of spiders and Lènnell ALLEN for operating the electron microscope. C.E.
GRISWOLD, P.S. Jackson, and VJD. Roth are gratefully acknowledged for helping
me locate spiders in the field. The assistance of the Southwestern Research Station of the
American Museum of Natural History is much appreciated. Thanks go to RJJ. DANIELS,
and T. MARSH for assistance with the manuscript. This work was supported in part by
N.S.F. grant number BMS 75-09915 to P.N. WITT.
% References
ANDERSON, JJfl 1974. — Responses to starvation in the spiders Lycosa lenta Hentz
and Filistata hibemalis (Hentz). — Ecology, 55: 576-585.
ALTMANN, SA., 1968. — Primates, in: Animal communication (ed.TA.SEBEOK).pp.
466-522. Indiana University Press, Bloomington.
BASTOCKjM., 1967. — Courtship: an ethological study.Aldine, Chicago.
BERLAND, J., 1916. — Note préliminaire sur le cribellum et le calamistrum des araignées
cribellates et sur les moeurs des araignées. — Arch. ZooL Expér. Gén., : 53-66.
BlLLAUDELLE, H., 1957. — Zur Biologie des Maurespinne Dictyna civica (H. Luc.)
(Dictynidae, Araneidae). — Z Angew. Entomol., 41 : 474-512.
BRISTOWE, W.S., 1958. — The world of spiders. Collins, London.
BRISTOWE, W.S. & LOCKET,- G .H., 1926. — The courtship of British lycosid spiders
and its probable significance. — Proc. ZooL Soc. London, 1926: 317-347.
BURGESS, W., 1976. — Social spiders. — Set Amer., 234 : 101-106.
CHAMBERLIN, R.V. & GERTSCH, WJ., 1958. — The spider family Dictynidae in
America north of Mexico. — Bütt. Amer. Mus. Nat. Hist., 116 : 1-152.
DANE, B. & van der KLOOT, W.G., 1965. — An analysis of the display of the goldeneye
duck (Bucephala clangula(L.)). — Behaviour, 22 : 282-328. ; T
Dig U ET, L., 1915. — Nouvelles observations sur le mosquero ou nid d’araignées sociales.
— Butt. Soc. Acclim. France, 62: 240-249.
DOBZHANSKY, T., 1970. — Genetics of the evolutionary process. Columbia University
Press, New York.
FOX, L.R., 1975. — Cannibalism in natural populations. —Ann. Rev. EcoL Syst, 6 : 87-
106.
GEIST, V., 1971. — Mountain sheep: a study in behavior and evolution. Chicago Univer-
sity Press, Chicago.
GERHARDT, U., 1924. — Weitere Studien über die Biolope der Spinnen. — Arch.
Nuturg., 90 : 85-192.
RJR. Jackson
GERHARDT, U, & KaESTNER, A., 1937. — Araneae, ih: Handbuch der Zoologie, vol 3
(ed. W.G. KüKENTHAL), pp. 394-656.DeGruyter, Berlin.
GREGG, M., 1961. — The mating of Ixeuticus longinuus. — Proc. Roy. ZooL Soc. New
South Wales, 1558-59 : 85-86.
GRISWOLD, CJE., 1977. — Biosystematics of Habronattus in California. M. Sc. Thesis,
University of California, Berkeley.
GWINNER-HaNKE, H., 1970. — Zum Verhalten zweire stridulierender Spinnen Steatoda
bipun eta ta Linné und Teutana grossa Koch(Theridiidae, Araneae), unter besonderer
B eruchsi ch tingling den Fortpflanzungverhaltens. — Z. TierpsychoL, 27: 649-678.
HAMILTON, WJD., 1964. — The genetical evolution of social behaviour.—/. Theoret
BioL, 7 : M6.
HARRISON, J.B., 1969. — Acoustic behaviour of a wolf spider, Lycosa gulosa. — Anim.
Behav,., 17: 14-16.
HEDIGER, H., 1950. — Wild animals in captivity. Butterworth, London.
HUNTINGFORD, F JV., 1976. — The relationship between inter- and intraspedfic aggres-
sion. — ^4nim. Behav., 24 : 485-497.
JACKSON, RJR., 1974.Rearing methods for spiders. —/. Arach, 2 : 53-56.
JACKSON, RJR., 1977a. — Courtship versatility in the jumping spider, Phidippus johnsoni
(Araneae: Saltiddae). ~~Amm. Behav., 25: 953-957.
JACKSON, R.R., 1977b. — An analysis of alternative mating tactics of the jumping
spider Phidippus johnsoni (Araneae, Saltiddae). — /. Arach., 5 : 185-230.
Jackson, 1977c. — Comparative studies of Dictyna and Mallos (Araneae, Dicty-
nidae) : III. Prey and predatory behavior. — Psyche (in press).
JACKSON, RJt., 1978a. — Comparative studies of Dictyna and Mallos (Araneae, Dicty-
nidae). I. Social organization and web characteristics. — Revue Arachnologique, 1
(4): 133-164.
JACKSON, RJR., 1978b. — Male mating strategies of dictynid spiders with differing types
of social organization. — Symp. ZooL Soc. London, 42: 74-88.
JACKSON, RJR., 1978c. — Web sharing by males and females of dictynid spiders. —Bull
British Arach Soc. (in press).
JACKSON, R.R., 1978d. — Predatory behavior of the social spider Mallos gregalis: Is it
cooperative? (in review).
JACKSON, R.R. & SMITH, S.E., 1978. — Aggressions of Mallos and Dictyna (Araneae,
Dictynidae): Population characteristics. — Psyche (in press).
KRAFFT, B., 1970. — Contribution â la biologie et à l’éthologie d’Agelena consociata
Denis (Araignée sodale du Gabon). Première partie. — BioL Gabon., 6: 199-301.
LEGENDRE, R., 1963. — L’audition et l’émission de sons chez les Aranéides. — Ann.
BioL, 2 : 371-390.
LOCKET, GJL, 1926. — Observations on the mating habits of some web-spinning spiders
with corroborative notes by W.S. Bristowe. — Proc. ZooL Soc. London, 2 : 1.125-
1.146.
LOHER, W. & CHANDRASHEKARAN, MX., 1972. – Communicative behavior of the
grasshopper Syrbula fuscovittata (Thomas) (Gomphocerinae) with particular consi-
deration of the male courtship. — Z TierpsychoL, 31: 78-97.
13!
MANNING, A., 1965. — Drosophila and the evolution of behaviour. — Viewpoints bt
Biology, 4: 125-169.
MANNING, a., 1966. — Sexual behaviour. — Symp. ZooL Sbc. London, 3 : 59-68. y
Maynard Smith, J. & Price, GJL, 1973.— The^ logic of animal conflict – Nature,
246: 15-18.
MAYR, E., 1963. — Animal species and evolution. Belknap, Cambridge, Mast
McKEOWN, K.C., 1936. — Spider wonders of Australia. Angus and Robertson, Sydney.
MONTGOMERY, T.H., 1903. — Studies on the habits of spiders, particularly those of
the mating period. — Proa Acad Nat Set Philadelphia, 55 : 59-149.
MORRIS, D.J., 1956. — The function and causation of courtship ceremonies. In: L’ins-
tinct dans le comportement des animaux et de fhomme (ed. P.P. GRASSÉ), pp.
261-287. Masson et Cie, Paris.
MOYER, K.E., 1968. — Kinds of aggression and their physiological basis. — Comrru
Behav. Biol, A, 2 : 65-87.
Otte, D., 1974. — Effects and functions in the evolution of signaling systems. — Ann.
} Rev. Ècol Syst, 5 : 385-417.
PECKHAM, G.W. & PeckhAM, E.G., 1889. — Observations on sexual selection in spiders
of the family Attidae. — Oca Pap. Wisconsin Nat Hist Soa, 1: 3-60.
PECKHAM, G.W. & PECKHAM, E.GJ 1890. — Additional observations on sexual selec-
tion in spiders of the family Attidae. — Oca Pap. Wisconsin Nat. Hist. Soa, 1:117-
151.
PLATNICK, N., 1971. — The evolution of courtship behaviour in spiders. — Bull British
Arach. Soa, 2 : 40-47.
Rhijn, J.G. van, 1973. — Behavioural dimorphism in male ruffs Philomachus pugnax (L.)
— Behaviour, 48: 153-229.
RICHARDS, O.W., 1927. — Sexual selection and allied problems in the insects. — Biol
Rev., 2 : 298-364.
ROBINSOfa, MR., 1975. — The evolution of predatory befikviour in araneid spiders, in:
Function and evolution in behaviour (eds. G. BAERENDS, C. BEER & A. MAN-
NING), pp. 292-312. Clarendon, Oxford
ROHLF, FJ. & SOKAL, R.R., 1969. — Statistical tables. Freeman, San Francisco.
ROVNER, J.S., 1978. — Adhesive hairs in spiders: behavioural functions and hydrauli-
cally mediated movement.— Symp. Zool Soa London (in press).
SAVORY, T.H., 1928. — The biology of spiders. Sidgwickand Jackson, London.
SHANNON, C.E. & WEAVER, W., 1949. — The mathematical theory of communication.
University of Illinois Press, Urbana.
SOKAL, R.R. & ROHLF, F.J., 1969. — Biometry: the principles and practice of statistics
in biological research. Freeman, San Francisco.
SPIETH, H.L., 1974. — Courtship behavior in Drosophila. — Ann. Rev. Entomol, 19:
385-405.
TURNBULL, A.L., 1973. — Ecology of the true spiders (Araneomorphae). — Ann. Rev.
Entomol, 18: 305-348.
WILSON, E.O., 1975. — Sociobiology: the new synthesis. Belknap, Cambridge, Mass.
132
R.R. Jackson
WITT, P.N., 1963. — Environment in relation to behavior of spiders. — Arch Environ.
Health 7 : 4-12.
WITT, P.N., 1975. — The web as a means of communication. — Bioscl Comm., 1: 7-23.
WRIGHT, S., I960. — Physiological genetics, ecology of populations, and natural selec-
tion. In: Evolution after Darwin (ed. S. Tax), pp. 429-470. University of Chicago
Press, Chicago.