Warning: Trying to access array offset on value of type null in /home/elisew5/public_html/drpeterwitt.com/wp-content/plugins/contextual-related-posts/includes/content.php on line 49
Images Collection
View this article in Search Friendly Plain Text
NOTE: This plain text article interpretation has been digitally created by OCR software to estimate the article text, to help both users and search engines find relevant article content. To read the actual article text, view or download the PDF above.
Reprinted tVom Journal of Morphology
Vol. No. 3, July 1975 © The Wistar Institute Press 1975
Post Embryonic Development of the Central Nervous
System of the Spider Argiope aurantia (Lucas)1
K. SASIRA BABU 2
North Carolina Department of Mental Health Division of Research, Raleigh,
North Carolina 27611
ABSTRACT Volumetric and histological changes of the central nervous system
were studied during post embryonic development of a spider, Argiope aurantia.
The neural mass of Argiope grows allometrically with respect to volume of the
céphalothorax and body weight. In the first instar 46% of the cephalothoracic
volume constitutes the neural mass and this is reduced to 4 9&^in the female (9th
stage) and 12% in the male (7th stage) spider.
Growth curves for the cephalic ganglion, measured at all stages, represent a
straight line. The neural mass of females is two and a half times larger than that
of the males. The ganglion increased 24 fold in female and 10 fold in male spiders.
Addition of neural mass occurs in all stages.
The brain volume is greater than that of the subesophageal ganglion in the first
two instars. In subsequent stadia, the subesophageal ganglion grows faster, and
in females it is finally three times and in males two times larger than the brain.
Growth of cortex and neuropile depict exponential curves. Comparison of
growth patterns of these shows an inverse relationship during development.
While the volume of the cortex ||| higher in the first two or three stages, the vol-
ume of the neuropile is higher in the remaining stadia. The causes for this growth
pattern are discussed.
Counts of cell numbers show that there is a constant population of neurons
throughout the post-embryonic development. The number of nerve cells in females
is higher than in males, 119$ in the subesophageal ganglion and 58% in the
brain.
The growth of the cortex is partly accomplished by an increase in cell volume.
In male and female spiders the increase in Type-B cells is 20 and 50 fold, while
that of large motor neurons is 200 and 600 fold respectively. The motor neurons
of 20 p and above number 63 in male and 916 in female adult spiders.
The growth of neuropile occurs through an increase of dendritic arborization
and axonal branching. The largest axons measure 1 p in the first and 16 p in
adult stages. An increase of incoming sensory fibers is also noticed during devel-
opment.
Invasion of neural lamella into cortex and neuropile increases during develop-
ment. Neural lamella which are 1—2 p in the first stage grow to 40—100 p thick-
ness in adult female spiders, near the origin of the main nerves. One type of astral
cells, counted in neuropile, increases 10 fold.
The appearance of a central body and the beginning of web construction co-
incide during the second instar. The relationship between these two is discussed.
Although the spiders have been used for
several kinds of experimental studies, the
post embryonic development of their central
nervous system has received little atten-
tion. However the nervous system of the
adult spider has been the subject of several
earlier investigations. Its external morphol-
ogy was described by Blanchard (’59), Po-
cock 002), Haller (12), Hilton (12), Bux-
ton (17), Gerhardt and Kaestner 037),
Millot 049), Legendre 053), Firstman 054)
and Babu 065, ’69). Its histology and
1 This paper is dedicated to the memory of Prof. K.
Pampapathi Rao on the first anniversary of his death.
2 Present address: Department of Zoology, Sri Ven-
kateswara University, Tirupati, Andhra Pradesh, India.
J. Morph., 146: 325-342.
325
K. SASIRA BABU
326
anatomy was reported by Saint-Rémy (’87).
Hanstrom (’19, ’21, ’28, ’35), Legendre
(’59), Babu (’65, ’69) and Meier (’67).
Studies on post embryonic development
of several groups of arthropods and partic-
ularly of insects have received considerable
attention. In spite of several attractive fea-
tures, similar studies on spiders are lack-
ing. Spiders pass through a long period of
post embryonic development in which the
pattern of growth can be measured. They
adapt quickly to the laboratory conditions
and this makes them easy to rear and study
at all stages. The amazing behavioral reper-
toire of spiders is a special attraction; its
understanding demands a study of the
growth pattern of the central nervous
system.
The aim of the present work is to study
in detail the factors that contribute to the
growth of the nervous system of female and
male spiders of Argiope aurantia. Keeping
this in view, attempts are made to study
the growth pattern of the central nervous
system in relation to body size, and the
inter-relationship between cortex and neu-
ropile, and also the organization and growth
of non-nervous glial elements in all the post
embryonic stages.
MATERIALS AND METHODS
Cocoons of the orb web spider Argiope
aurantia Lucas (family: Araneidae, Levi,
’68) were collected at Raleigh, North Car-
olina in the field in the months of December
and January. In February and March the
spider lings were released into cages by cut-
ting open the cocoons. Each animal from
the second stage (“Stage or stadium or in-
star,” refers to an intermolt period; i.e.,
seventh stage after sixth molt) was kept
separately in a glass jar and data sheets of
the sex, age and time of molting for each
animal were maintained. The younger ones,
up to the fifth or sixth stage were fed with
gnats and the older ones with house flies.
The first stage was brief (approximately
two days), and the young ones underwent
their first molt within the cocoon. The
total number of molts from females varied
from seven to nine whereas the number of
molts for males varied from five to seven.
Twenty-five percent of the population were
males. The females became gravid and laid
cocoons after the eighth molt. They died
normally about four to six weeks after lay-
ing the last of one to three cocoons. This
period is referred to in the text as post co-
coon. At an early age, male and female
Argiope look alike, feed alike and build
alike, as do Araneus diadematus (Witt,
>71). ■mKk
At the fourth stage in the male, external
differentiation of sex organs occurs, when
the terminal pedipalpal segment begins to
swell. Hence descriptions of the central
nervous system for males are given only
from fourth to seventh stage.
The male and female spiders were
weighed before fixation. The chelicerae,
legs and abdomen were cut off, and the
céphalothorax was fixed in Zenker’s, Hel-
ly’s, or Bouin’s. Granular “Histowax of
M.P 56—58°C was used throughout. Sec-
tions of the entire céphalothorax were cut
at 8—10 /A in the three cardinal planes; very
good paraffin ribbons, even of the cuticle
were obtained. Some of the serial sections
were stained in Palmgren (’48) silver tech-
nique and others in basic stains like Azan,
Heidenhain’s haematoxylin and Mallory’s
phosphotungstic haematoxylin.
Outlines of the entire céphalothorax,
the central nervous system, the neuropile
and cortex were made with a camera luci-
da at a known magnification on millimeter
graph sheets. The areas thus enclosed were
counted and actual volumes were ob-
tained by correcting for the magnification
of these outlines and for the thickness of
the sections. These operations gave the vol-
umes of thglentire céphalothorax, the ce-
phalic nerve mass, the volumes of the
cortex and neuropile for supra and sub-
esophageal ganglia. These volume mea-
surements were made for all the stages in
male and female spiders.
The total nerve cell estimates in the
nerve mass were obtained in the following
way: nuclei were counted in every alter-
nate section and the total multiplied by
two gave the crude count of total number
of cells. The correction factor suggested by
Marrable (’62) has been applied to these
cell counts. Nuclei were counted at a mag-
nification of 400 X , and a grid was insert-
ed into one eyepiece to facilitate counting.
The cell and nuclear diameters were mea-
sured with an oil immersion lens. All cells,
glial and neural, were counted in the cor-
tex because of the difficulty in distinguish-
ing one from the other.
GROWTH OF THE SPIDER NERVOUS SYSTEM
327
TABLE 1
Mean volume and standard deviation of the central nervous system and céphalothorax of female and mean
body weight of female and male spiders (N = 4) A. aurantia. The volumetric data are given in
mm3 X 10 ~10
S 1 S 2 S 3 S 4 S 5 S 6 S 7 S 8 S 9 PC
C eph alothor ax 2930 3490 11350 16619 33350 59473 121726 181998 541222 * t
± ± ± ± ±_ Hh
90.07 384.3 492.3 5139 3860 9936 7236 17010 35550
Nerve mass 1170 1659 4262 5378 7573 9337 11782 14741 ’T9430 24523
± ± IS ± ± + ±
47.80 32.72 419.8 957.2 464.5 561.4 970.1 825.0 737.2 1134.0
Cortex 937 1001 1355 2472 2442 3255 3887 4159 5309 6190
+ ± ■a i ± ± ± ± ±
33.86 38.77 53.04 10.98 72.85 272.5 467.3 182.7 714.5 595.4
Fibrous mass 243 695 2669 3079 4922 6166 7680 9638 13822 18280
± ± ± ± ■ ± ±
25.48 28.87 414.2 493.9 309.0 299.1 278.6 803.5 814.1 748.5
Female body 0.4 0.4 1.0 2.5 4.0 10.0 26.0 82.0 140.0 220.0
weight (mg) ± ±
0.3 0.75 1.08 4.03 8.91 36.37 52.26 50.43
Male body 2.0 4.0 7.0 10.2
weight (mg) + Î
0.46 0.96 1.85 4.5
Sl-9 represent the number of stadia. PC: Post cocoon-laying period.
1 Even though the whole céphalothorax was sectioned during this period, parts of it were removed for easy penetration of
the fixative. Hence data on volume measurements PC could not be furnished.
RESULTS
The post embryonic development of the
spider resembles that of heterometabolous
insects like cockroaches, crickets, and lo-
custs. The hatchling resembles an adult
spider in all physical aspects except size.
In Argiope, reared under laboratory condi-
tions, the length of intermolt periods varies
from 11 to 15 days in females (Babu, ’73)
and from 15 to 17 days in males. The spi-
ders are active during the entire postnatal
period and increase in size and weight after
each molt. Thus spiders lack a quiescent
period like that of holometabolous insects.
The total body growth in female and
male spiders shows sexual dimorphism
(table 1). There is no change in body weight
in the first and second instars (0.4 mg).
From third to sixth stage the body increases
from 1.0 to 10.0 mg in female spiders. A
spurt in body growth from 26.0 to 220.0
mg occurs from the seventh to the post co-
coon period. Gravid females weighed 297.0
± 11.09 mg.
The body weight of female and male
spiders are nearly equal in the fourth and
fifth stages. The growth rate in males, how-
ever, slows down as they attain maturity.
The body weights of males are approxi-
mately one third of those of the females in
the seventh stage. Such differences have
been reported previously for the body
weights of spiders, Araneus diadematus,
(Witt et al., 72).
A characteristic feature of the spider
central nervous system (CNS) is the high
degree of cephalization. There is a single
large fused cephalothoracic nerve mass at
the anterior end of the animal (Babu, ’65).
This compound neural mass consists of a
dorsally located supraesophageal ganglion
or the brain, and a ventrally placed sub-
esophageal mass (SEG). The latter includes
a pair of pedipalpal and four pairs of leg
ganglionic masses and several pairs of
small abdominal ganglia.
The CNS of spiders conforms to the typ-
ical arthropod plan. It consists of a cellular
cortex surrounding a highly complex fi-
brous mass or neuropile (Bullock and
Horridge, ’65). The neuropile forms the
most important structure in invertebrates
because this is the only known place of
neuronal contacts where the process of
functional integration takes place.
Relationship between growth of
céphalothorax and central
nervous system
The cephalized ganglionic mass occupies
K. SASIRA BABU
46% of the entire céphalothorax in the
hatchling. At each successive stage the per-
centage of nerve mass in the céphalothorax
diminishes gradually. In the ninth stage
only 4% of the céphalothorax constitutes
the CNS.
Figures 1 and 2 represent growth curves
of céphalothorax and nerve mass. The CNS
shows a continuous but slow growth rate at
all stages. More than 75% of the volume is
added to the céphalothorax during the sev-
enth and subsequent stages. The total body
weight grows at a similar rate during de-
velopmental stages.
Fig. 1 Graphic representation of growth in vol-
ume of céphalothorax (solid lines) and total nerve
mass (dotted lines) during post embryonic devel-
opment in male (short lines) and female (long lines)
Argiope aurantia: ordinate, volume in logarithm of
mm3; abscissa: stage in development.
3
3 4 5
Fig. 2 Growth relationships between cephalo-
thoraric mass, plotted as logarithm of volume in
mm3 on the ordinate, and total nerve mass, plotted
in the same way on the abscissa. Full circles repre-
sent mean values for females, and the solid line
indicates overall growth relationships for females;
open circles and dotted line show comparable re-
lationships for males.
Fig. 3 Semi-logarithmic plot of volumetric
growth, separately for total nerve mass, fibrous
mass, and cortex in female Argiope aurantia spi-
ders during post embryonic development. Note the
reversal in growth trend between cortex and fibrous
mass during earlier stages.
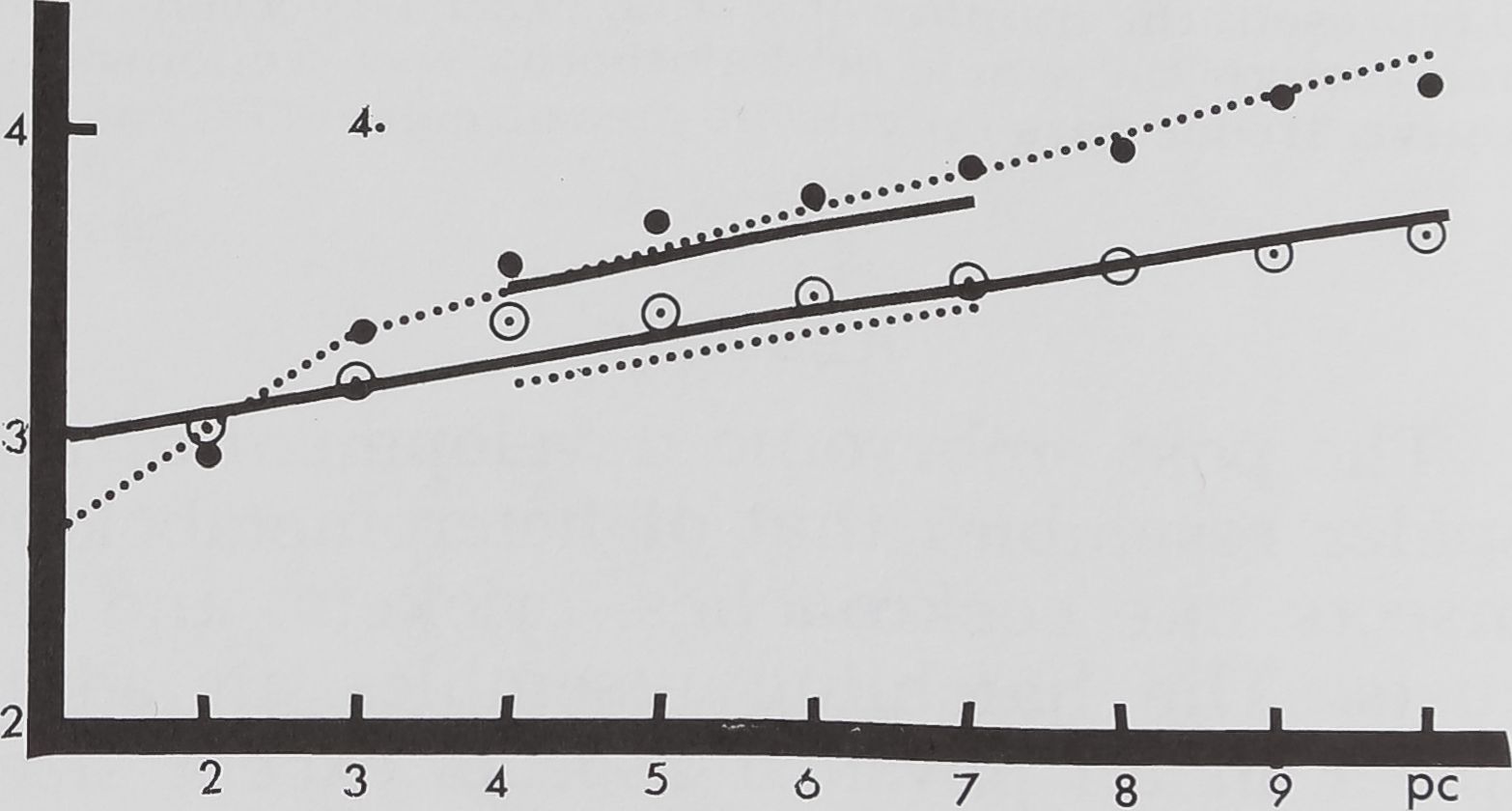
KSpg. 4 Growth of total volume of brain (solid
lines) and sub esophageal ÇSEG) nerve mass (dot-
ted lines) in male (short lines) and female (long
lines) spiders, plotted in log mm* 3 on the ordinate
vs developmental stage on the abscissa. For females
the means to which the lines have been fitted are
shown as open circles for SEG and full circles for
brahtt volume; the means for males he all on each of
the two short lines.
Growth curves are slightly flatter for
male than for female spiders. In the sev-
enth stage the total volume of the céphalo-
thorax of male spiders forms 70% of that of
female spiders. And the total nerve mass of
males is slightly smaller than that of female
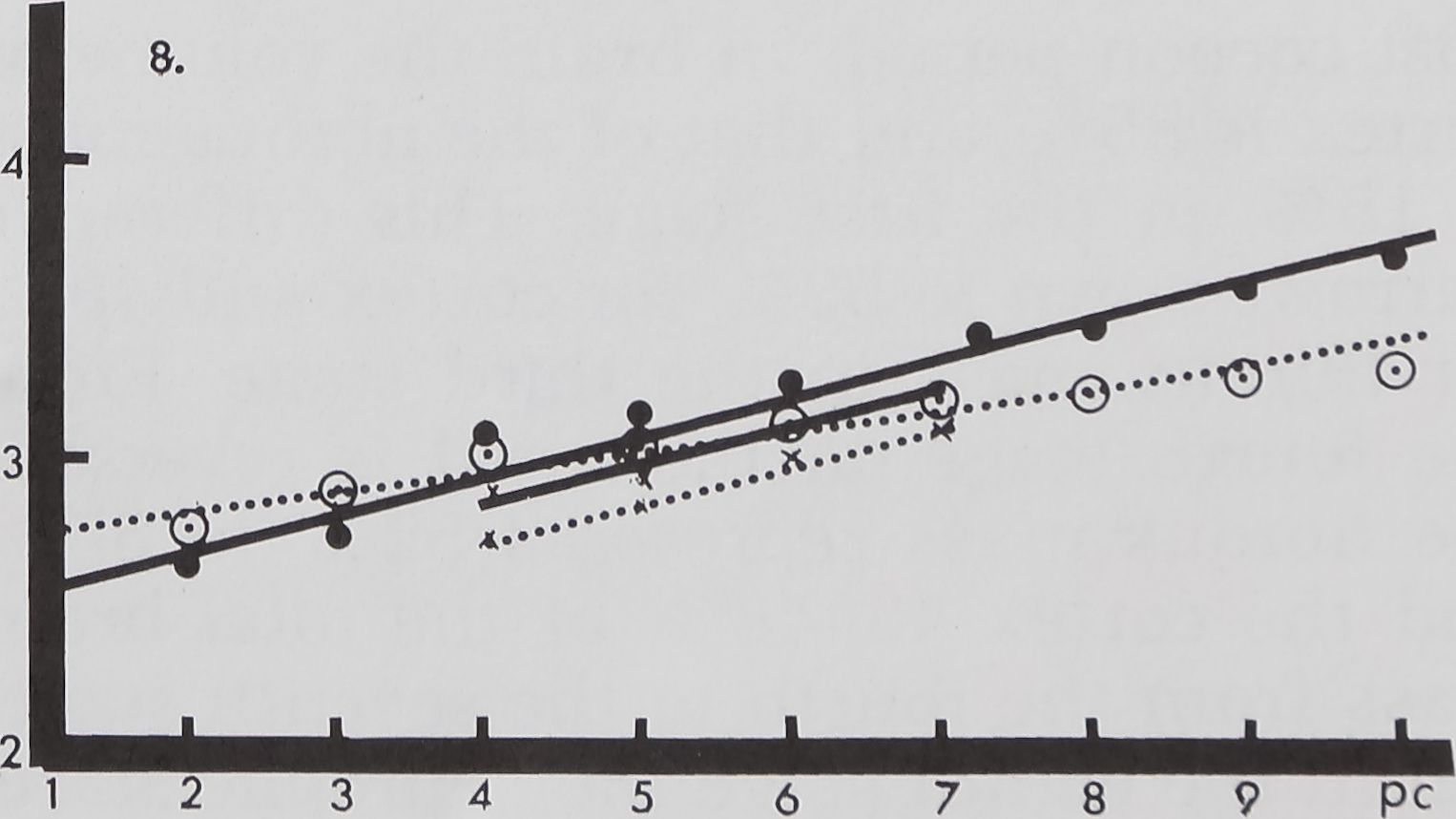
spiders at the seventh stage (fig. 1).
Growth of the total volume of the
ganglionic mass
The total mass of CNS grows in such a
way that the curve derived from measure-
ments of all stages in female and male spi-
ders forms an almost straight line (figs.
1, 3) when plotted on a logarithmic scale.
GROWTH OF THE SPIDER NERVOUS SYSTEM
329
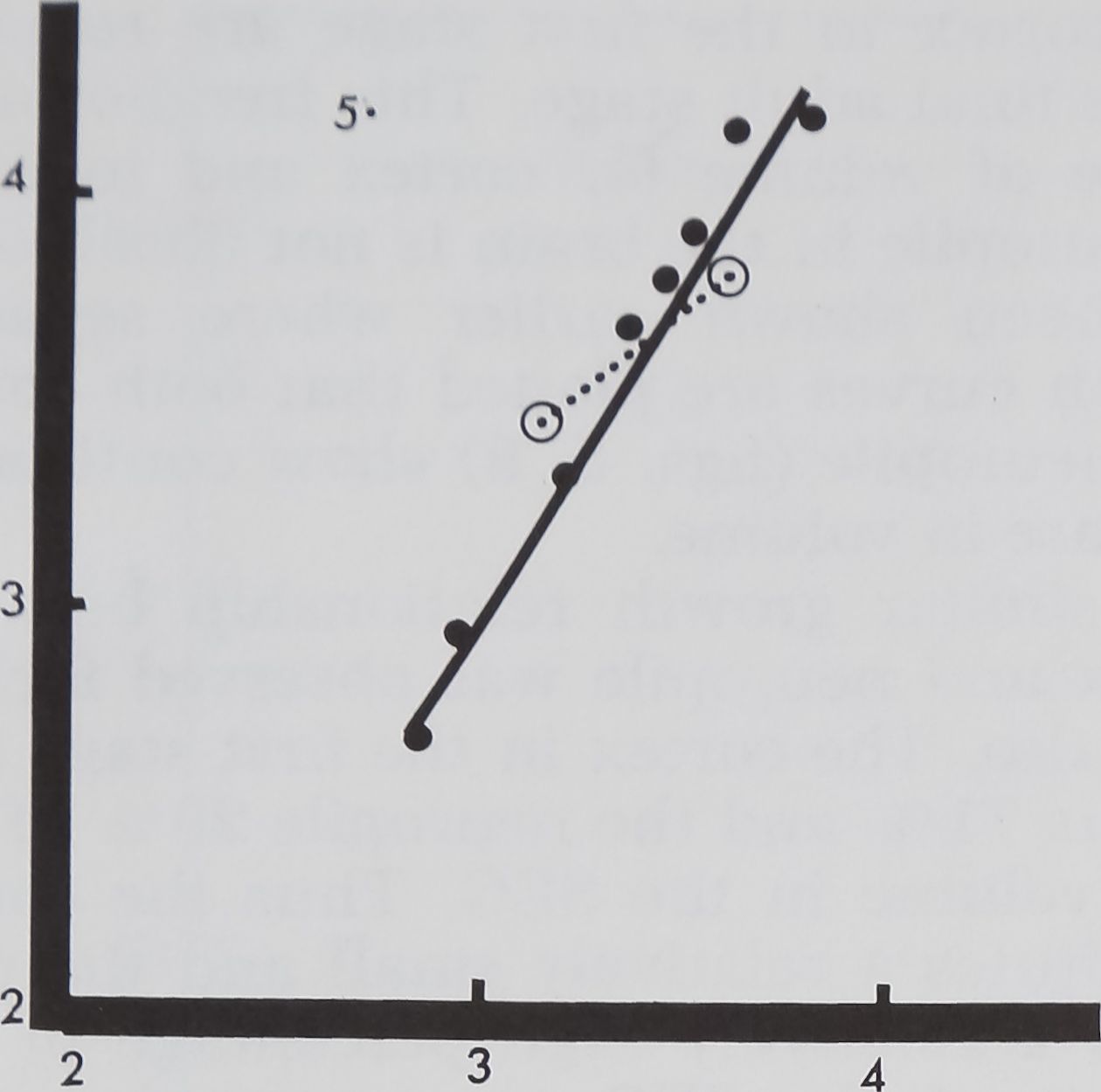
Fig. 5 Relative growth in volume of total SEG
(on ordinate in log mm3) as compared to total brain
volume (abscissa in log mm3) for males, dotted line
and open circles, and females, sobd line and full
circles.
23456 789 PC
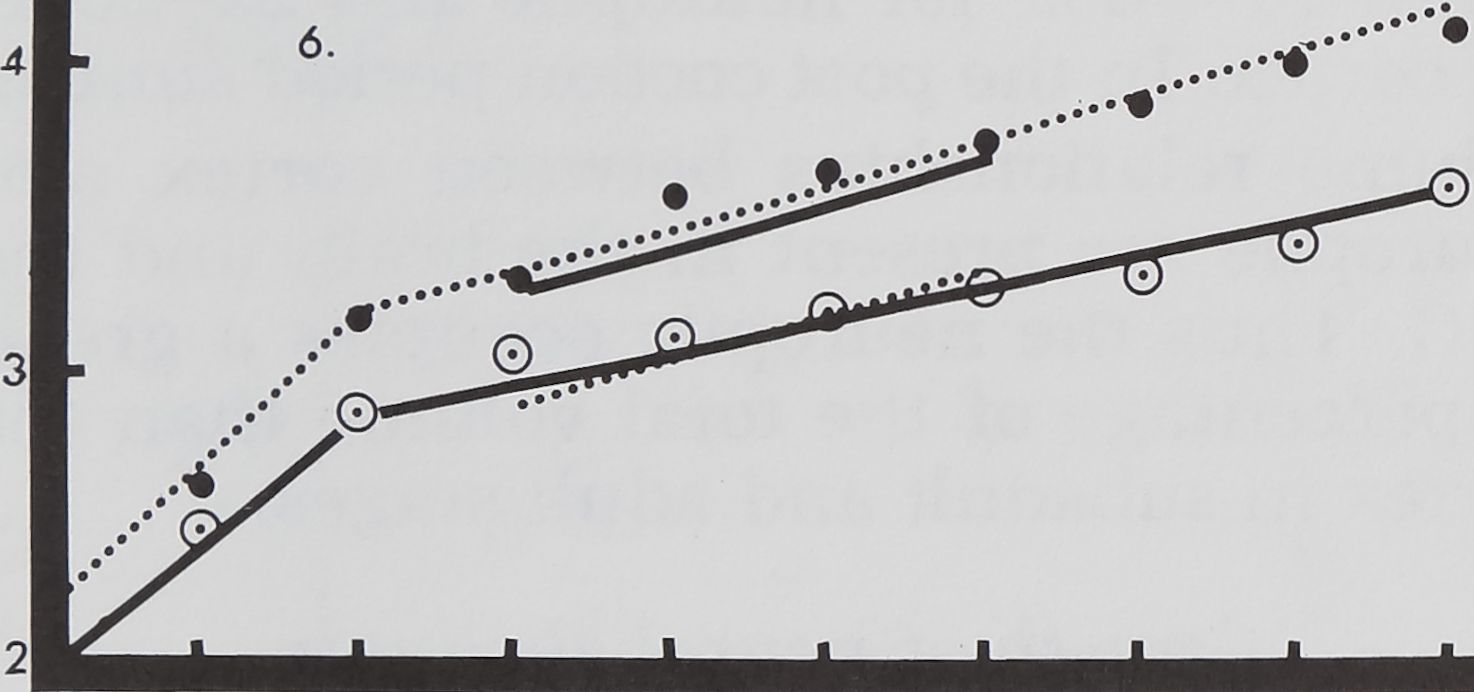
Fig. 6 This plot shows the volumes of the fi-
brous mass of brain (solid lines) and SEG (dotted
lines) for male and female spiders during postem-
bryonic development. Only the mean values for
females are indicated.
The central nervous mass increases in
volume during each larval stage. It reaches
50% of the total volume by the seventh
stage. During the subsequent two molts
and in the post cocoon period, the remain-
ing 50% growth is reached. There is addi-
tion of neural mass at each successive
stage, but maximal addition occurs in the
last stages. The growth rate of the neural
mass in males is slightly lower than that
of the females at corresponding stages.
Figure 4 shows separate growth curves
for brain and SEG. These ganglia show an
increase in volume at each stage. The sub-
esophageal nerve mass in females grows
faster than the brain mass. Both reach
50% of the adult volume by the seventh
stage. It is interesting to note that in the
first and second stages the brain volume is
greater than the subesophageal mass. But
this trend is reversed in the third and sub-
sequent stages when the SEG grows faster
than the brain. In the seventh stage the
SEG and the brain volume reach 50% of
the adult neural mass. But the rate of
growth differs. In the seventh stage the
total brain volume is less than half of the
subesophageal mass. These volume differ-
ences further increase in the post cocoon
period, when the brain is only one third of
the SEG (fig. 5).
Growth of the neuropile and cortex
Volumetric measurements of the fibrous
mass reveal that its growth curve in brain
and SEG shows an exponential curve (fig.
6). In the later stadia the rate of growth of
the fibrous mass in the SEG is much great-
er than that of the brain.
The fibrous mass in the brain also in-
creases rapidly in the last stages, but not
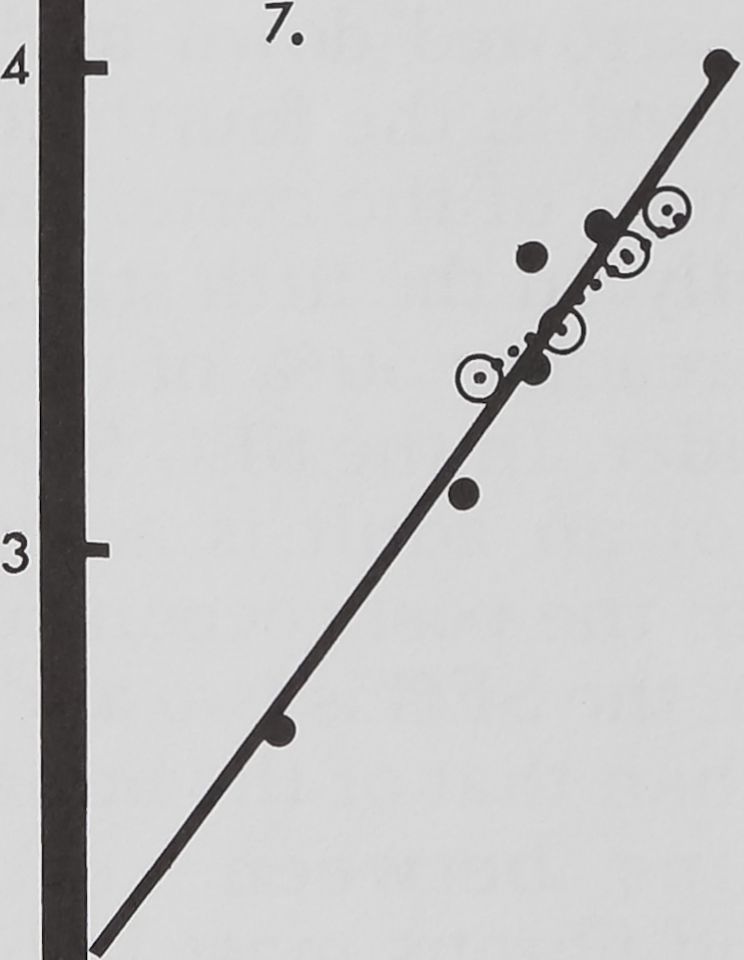
2 3 4
Fig. 7 Relative growth of fibrous mass of SEG
and brain, plotted in the same way as in figure 5.
Fig. 8 Growth curve for the cortex in the SEG
(solid lines and solid circles) and brain (dotted lines
and open circles or X’s) plotted in log volume mm3
on the ordinate vs developmental stage on the ab-
scissa. The shorter lines represent male growth
data.
K. SASIRA BABU
330
at the same rate as that of the SEG (fig. 7).
In the eighth stage the volume of the fi-
brous mass has reached half of the mass of
an adult brain. But 50% volume of the
fibrous mass in the SEG is reached in the
seventh stage. In hatchlings the volume of
fibrous mass in the SEG and the brain is
nearly equal. But during development addi-
tion of fibrous mass to the SEG occurs at a
higher rate than to the brain. In the third
and fourth stages the rate of addition of
fibrous mass in the SEG is twice that of
the brain and in subsequent stages it reach-
es nearly three times.
Volumetric measurements of the cortex
also show a linear growth rate (fig. 8). The
volume of cortex in the brain and SEG
continues to increase through all stadia. An
interesting aspect of cortex growth is cross-
ing of the curves at an early stage. Up to
the third stage, the cortex volume is great-
er in the brain than in the SEG. In the
hatchling the cortex in the brain has twice
the volume of that of the SEG. This differ-
ence is gradually narrowed down and the
relative size is reversed in the fourth stage,
after which the volume of the cortex in the
SEG increases rapidly. In the fifth stage the
cortex in the brain reaches 50 % of the vol-
ume of an adult spider. In the SEG 50% of
the cortex growth of an adult is achieved
at the sixth stage. In the post cocoon stage,
the cortex volume in the SEG is two and one
half times greater than that of the brain.
The relationships between relative
growth of cortex and fibrous mass in brain
and SEG are a notable feature (figs. 6, 8).
In supra and subesophageal ganglia the
growth is exponential in early stages. In
later stadia the rate of growth slows down,
but volume continues to increase up to the
post cocoon period. In brain the volume of
cortex is 85% and that of the fibrous mass
is 15% in the first stage. This difference
narrows down to 52% for cortex and 48%
for fibrous mass in the third stage. From
the fourth stage on the trend is reversed:
the fibrous mass represents 52% to 55%
and the cortex 45-48% of the total brain
mass from the fourth to the seventh stage.
But in the eighth stage the neuropile starts
to increase again, while the relative vol-
ume of the cortex decreases. Post-cocoon,
the neuropile represents 73% and the cor-
tex 27% of the total volume of the brain.
Thus the relative percentages of neuropile
and cortex in the first stage are reversed
in the final adult stage. This trend of a de-
crease of volume for cortex and increase
in neuropile in the brain is not absolute: it
has been shown earlier where separate
growth curves are plotted that both cortex
and neuropile (figs. 5, 8) show continuous
increase in volume.
A similar growth relationship between
cortex and neuropile was observed for the
SEG also. The cortex in the first stage rep-
resents 71% and the neuropile 29% of the
total volume in the SEG. Thus the cortex
constitutes a relatively small and the neu-
ropile a relatively high percentage of the
volume of the SEG as compared to the
brain. Unlike in the brain the reversal trend
in growth rate of neuropile and cortex takes
place in the third stage. From the third to
the post cocoon period, a nearly uniform
growth relationship is maintained between
cortex and neuropile. This ratio varies be-
tween 71—75% for neuropile and 29—25%
for cortex. In the post cocoon period similar
volume relationships between cortex and
neuropile are present in the brain and the
SEG. Thus the neuropile occupies a great-
er percentage of the total volume than the
cortex in subadult and adult stages.
Growth of neural elements
Growth of cortex
The characteristic internal differentia-
tion of the CNS into peripheral cortex and
central fibrous mass is present from the
hatchling stage. This trend becomes more
marked in the subsequent stadia.
On the basis of size, staining and size of
axon, three different types of cells were re-
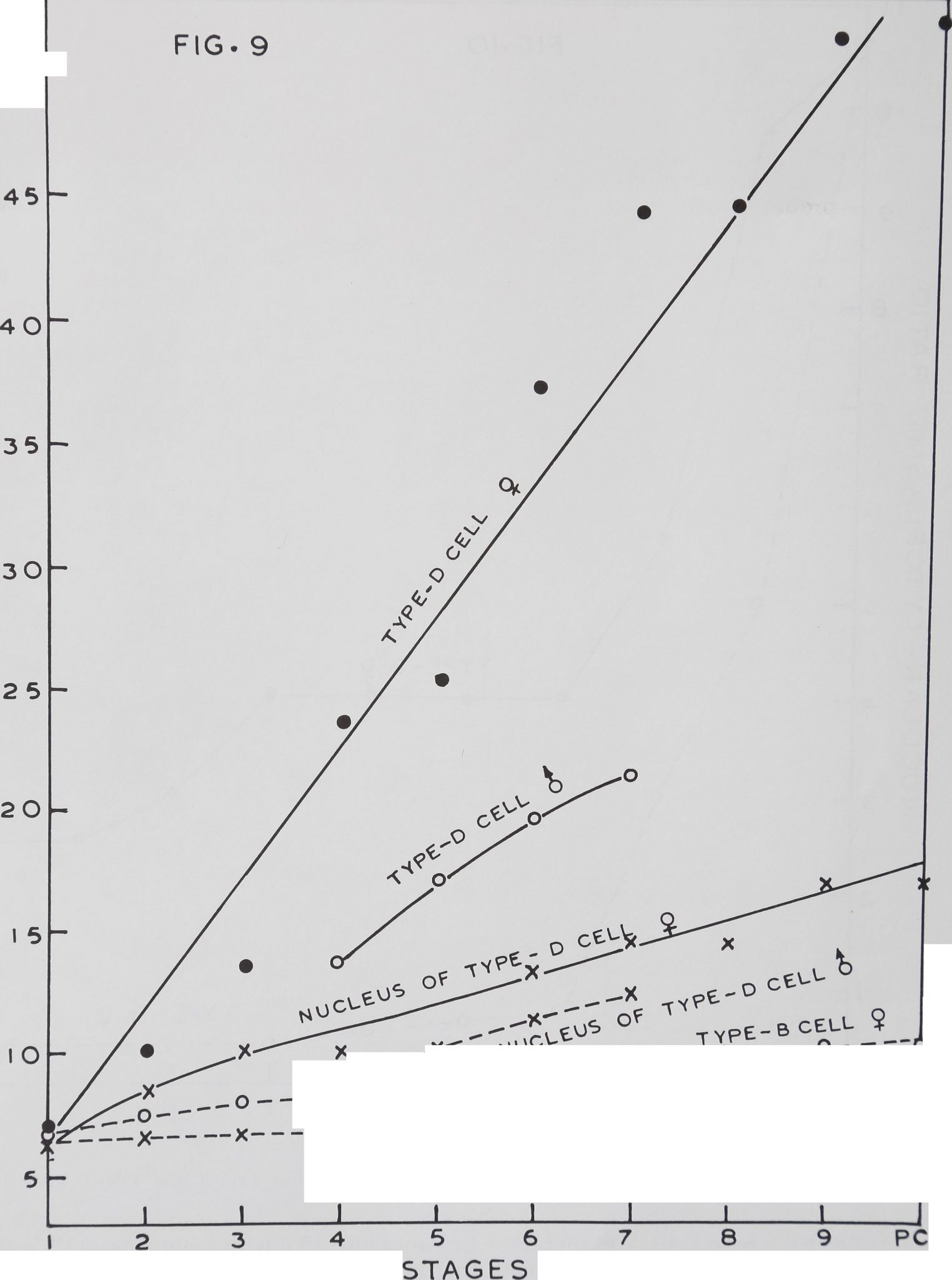
ported (Babu, ’65). The globuli type which
were classified earlier as Type-A cells are
absent. The Type-B cells in the first stage,
which measure 7.0 n with a nucleus of 6.4
/x, grow to a maximum of 10.5 fi with a
nucleus of 7.5 /u, in the post cocoon period
(fig. 9). Thus the Type-B cells show 50%
growth in volume from the first to the last
stage.
The Type-C are the neurosecretory cells.
It was reported that these cells also in-
crease in size from the second to the post
cocoon period (Babu, 73).
The Type-D, which are mostly motor
neurons (Babu, *65, *69), show continuous
growth from the first to the post cocoon pe-
DIAMETER OF NERVE CELLS IN MICRONS
GROWTH OF THE SPIDER NERVOUS SYSTEM
331
50 –
■ ■29
6–—O’ ^ T YPE-B cELL g_x-
■X x——X— “nucleus of
X____X—-X-X * TYPE- B CELLÇ
%______^————-‘ I
NUCLEUS OF TYPE-B CELL Ô
Fig. 9 The growth of Type-B and Type-D nerve cells in male and female spiders at various develop*
mental stages.
K. SASIRA BABU
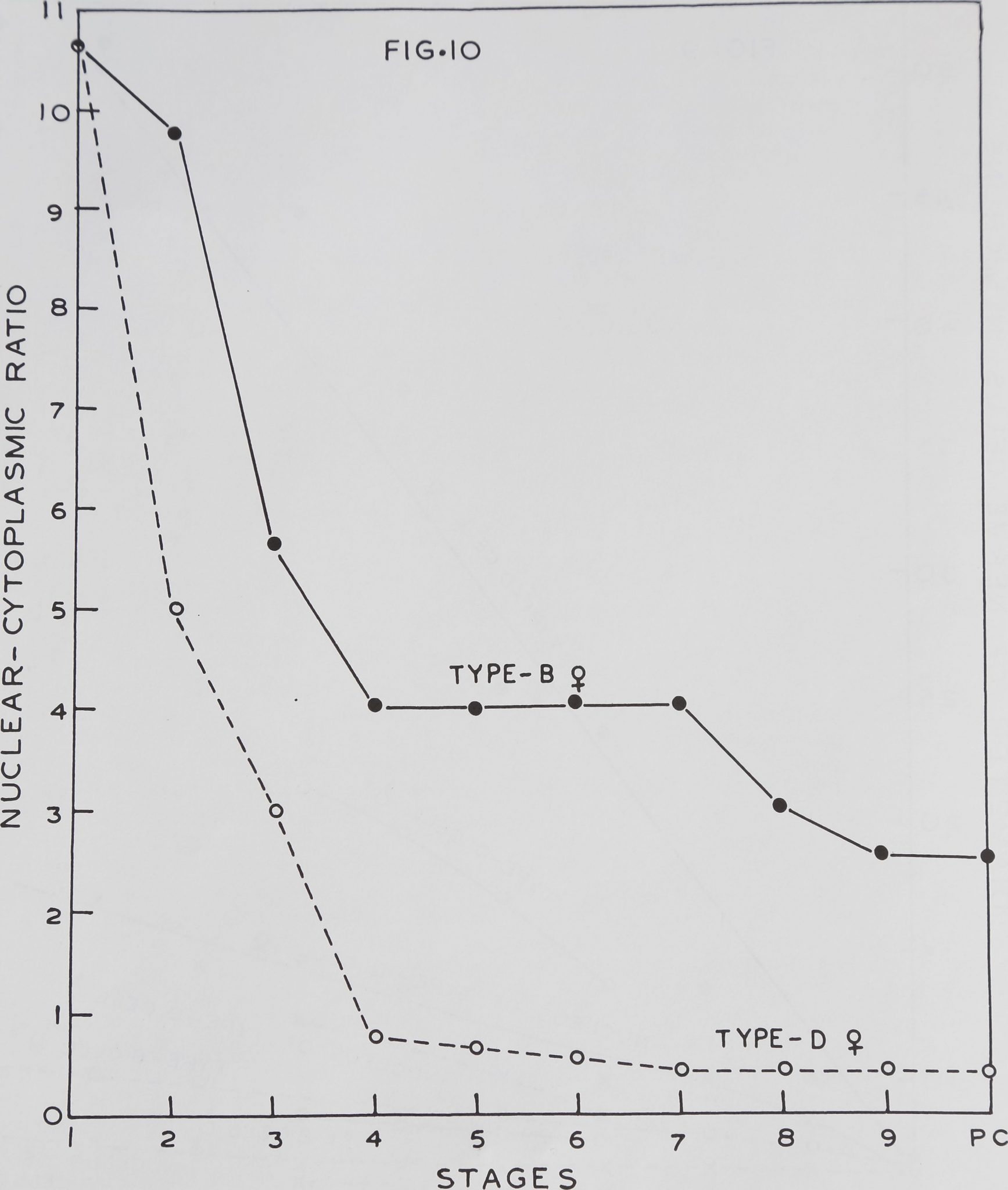
Fig. 10 Graph showing changes in nuclear-cytoplasmic ratios of Type-B and Type-D cells during
postembryonic development of female spiders.
riod (fig. 9). These cells are abundant in
all ganglia of the subesophageal mass. In
the first stage, the average cell diameter is
7.0 /Lt with a nucleus of 6.4 /*. Throughout
the post cocoon period, the largest cells
measure 55 /i with a nucleus of 17 show-
ing a 600% increase in volume.
The Type-B cell diameters in males re-
main at the same level from the fourth to
the seventh stage. But the growth pattern is
GROWTH OF THE SPIDER NERVOUS SYSTEM
333
different for Type-D cells. In the fourth
stage the Type-D cells of females measured
24 /it and that of the males measured 13
In the seventh stage their diameters were
45 /x and 21 for female and male spiders
respectively. Thus the largest motor neu-
rons of a female in the seventh stage are
twice the size of those of a male spider.
Figure 10 depicts the nuclear-cytoplas-
mic ratios of female spiders. The Type-B
and D cells have a high ratio (10.6) in the
first stage. This is reduced to 4.0 in Type-B
cells in the fourth stage and is maintained
at this level up to the seventh instar. But
in subsequent stages the ratio falls grad-
ually and reaches the minimum of 2.5 in
the last stadia and post cocoon period. The
nuclear-cytoplasmic relationship for Type-D
cells shows a greater decline from early to
later stadia. The initial ratio of 10.6 drops
to a mere 0.73 in the fourth stage. After
the fourth stage the ratio declines slowly
and reaches a steady level of 0.4 in the sev-
enth stage.
Thus nerve cell growth takes place most-
ly by an increase in cytoplasmic volume of
the cell. The Type-B cells show a small
growth rate and maintain a high nuclear-
cytoplasmic ratio. But in Type-D cells there
is an enormous increase of the cytoplasmic
volume without a concomitant growth of
the nucleus. Thus these cells are more plas-
matic and bulbous, unlike the more chro-
matic Type-B cells.
In the first stage there is no differentia-
tion of nerve cells into different categories
in supra and subesophageal ganglia. From
the second stage on the Type-B, Type-C and
Type-D cells are differentiated. A greater
percentage of nerve cells are Type-B, in
the brain and SEG. The Type-D cells are
confined to the SEG mostly, except a few
cells in the tritocerebral part of the brain.
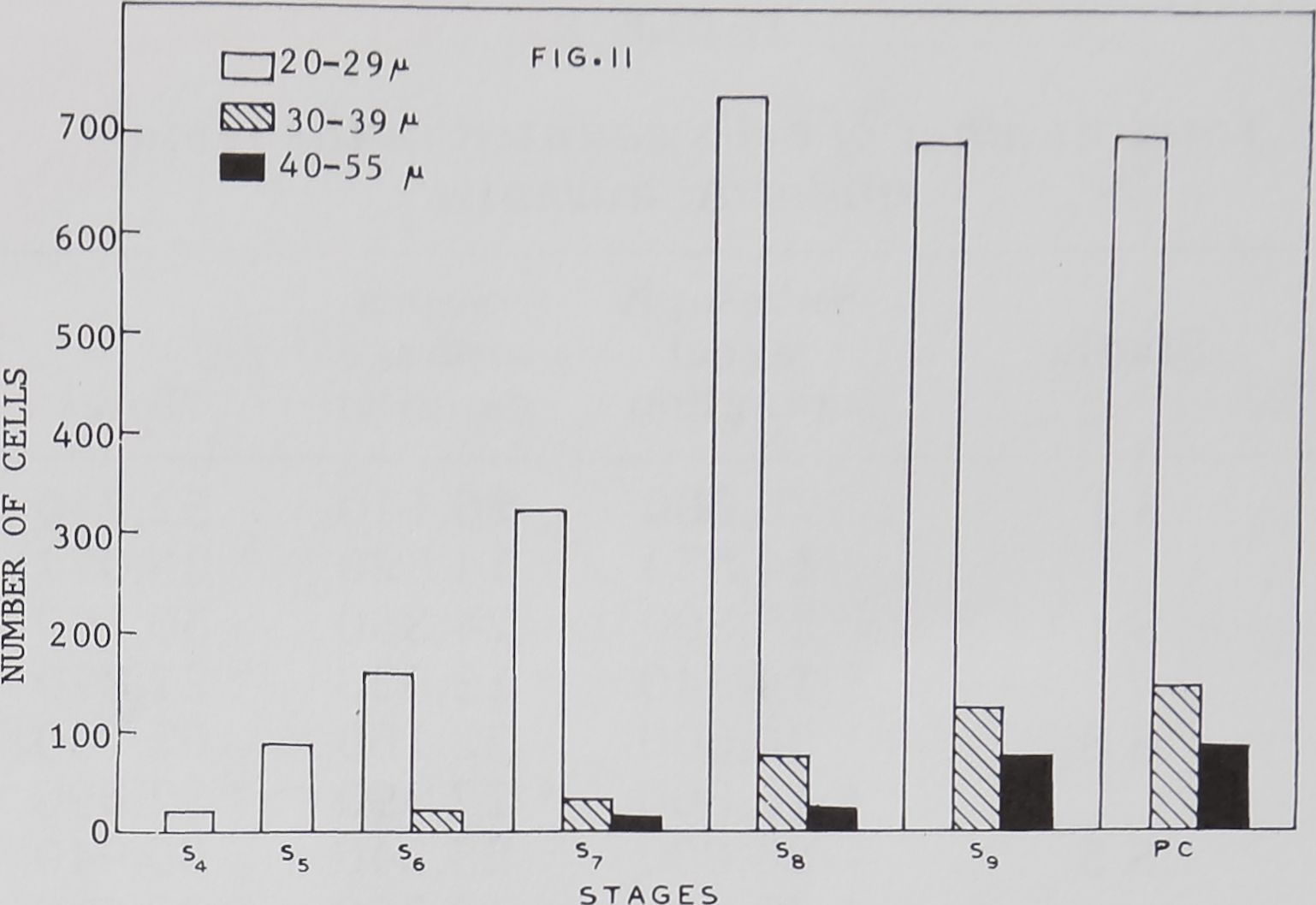
The increase in volume of cellular cor-
tex is dependent on the growth of cell size
and also on the number of large cells in
each stadium. The histogram in figure 11
depicts this aspect of growth. In the post
cocoon period, cells above 20 /x form 4.0%
of the total number of nerve cells in the ce-
phalic mass.
The cell volume and cell number in-
creases at each higher stadium. The 20—
30 size group cells appear first in the
fourth stage. Their number doubles in sub-
Fig. 11 The large motor neurons were arbi-
trarily classified into three groups based on their
diameter (20-29 /x; 30—30 /x and 40-55 /x). The
number of cells for each range were counted in all
stadia. The nerve cells of 20—29 /x range were ab-
sent in the first, second and third instars. The his-
togram shows a progressive increase in volume and
cell number of different diameters from the fourth
to the post cocoon period. The numbers represent-
ed in the histogram are from one count.
sequent stages and reaches a maximum
level in the eighth and subsequent stages.
From a mere 20 cells in the fourth stage,
they increase to a maximum number of
690—724 cells in the last stages.
The next size range of 30—40 ^ diam-
eter cells is found beginning with the sixth
stage. The number increases from 20 cells
in the sixth stage to a maximum of 140
cells throughout the post cocoon period.
Unlike the earlier described group of small-
er cells, these cells show a continuous in-
crease in number up to the last post co-
coon period. The increase in the number of
cells from one stage to the next is one and
a half to two times over that of the earlier
period.
The largest cells of 40—55 /x diameter ap-
pear in the seventh stage. These cells also
increase in number at each higher stadi-
um. The cells increase from 10 in the sev-
enth stage to a maximum of 76 in the post
cocoon period. In male spiders the differen-
tiation of cells is poor. In the seventh stage
there are only 63 of the largest cells in the
20—30 ix range.
Thus, the growth of the cortex takes
place by an increase in cell volume as well
as by an increase in the number of large
cells.
The total nerve cell population was
counted in female and male spiders. Tables
2 and 3 show that there is no increase in
K. SASIRA BABU
TABLE 2
Total number of cells counted in the female
spider A. aurantia
Stadia Subesoph- ageal ganglion Supra esophageal ganglion Total
S 1 26,600 26,140 52,740
* 14,554 * 14,520 * 29,074
S 1 25,800 24,360 50,160
* 14,340 * 13,530 * 27,870
S 3 30,600 32,160 62,760
* 15,300 * 17,390 * 32,690
S 5 25,600 25,340 50,940
* 12,800 * 13,700 * 26,500
S 6 28,320 22,400 50,720
* 14,160 * 13,333 *. 27,493
S 7 25,780 27,500 53,280
* 12,575 * 16,310 * 28,885
S 9 26,616 28,760 55,376
* 12,674 * 16,433 * 29,107
PC 26,540 30,140 56,680
* 12,640 * 17,223 * 29,863
P C 26,600 27,180 53,780
* 12,666 * 15,530 * 28,196
Mean value and 26,838 27,110 54,048
standard
deviation 1,498 2,818 3,694
* 13,623 * 15,329 * 28,853
997 1,512 1,655
The top numbers represent approximate counts of
cells. The totals with an asterisk represent numbers
after adjusting the correction factor.
SI—9, represents the number of stadia.
PC, post cocoon period.
total number of cells as the animal grows
from young to an adult spider. Division of
nerve cells was not noticed with the stain-
ing techniques used in the first stages, nor
at later stages. Thus by the time an em-
bryo is hatched the total number of nerve
cells is fully formed. From the data, it is to
be inferred that new nerve cells are not
added during post embryonic development.
The total number of cells in the brain is
only 13% more than in the SEG of a fe-
male spider. This higher number of nerve
cells occurs in spite of significant differ-
ences in the number of neurons and volume
of brain and SEG. The clue lies in cell size
and in their compact arrangement.
In the cephalic nerve mass of the male,
nuclear counts show that the total number
of nerve cells is smaller than in the female.
This is not surprising because the total
nerve mass and the volumes of the cortex
and fibrillar mass are consistently smaller
than those of females from the fourth to
TABLE 3
Total number of cells counted in the male
spider A. aurantia
Stadia Subesoph- ageal ganglion Supra- esophageal ganglion Total
SM 4 24,240 16,280 40,520
* 12,528 * . 9,303 * 21,831
SM 5 23,480 15,100 38,580
* 10,734 | 8,630 * 19,364
SM 5 23,380 17,520 40,900
* 11,690 * 9,344 * 21,034
SM 7 24,780 18,100 42,880
* 13,765 * 10,600 * 24,365
Mean value and 23,970 16,750 40,720
standard ±
deviation 572 1,156 1,526
* 12,179 * 9,469 * 21,648
±
1,114 871 1,803
The top numbers represent approximate counts of
cells. The totals with an asterisk represent number after
adjusting the correction factor.
SM 4—7, represent the number of stadia.
the seventh stage. The same trend in nu-
clear population is reflected here.
Growth of fibrous mass
The increase in cell size with each higher
stadium goes together with a corresponding
increase in axon diameter. Measurements
close to the cell body or on axons immedi-*
ately after they enter the neuropile gave the
following values:
In the first stage the largest fibre mea-
sured is 1 /Lt. This increases to 4 p in the
third, and to 9 p, in the seventh stage. In
the ninth and other post cocoon periods the
largest axon has a diameter of 16 /*. The
large axons arise from the large motor cells.
In adult males such large axons measuring
8 fi are comparable to those of the female
in the seventh stage. But such large axons
are few in number.
Observations of serial sections in the
three planes from early to late stages re-
veled that the various processes of a neuron
grow considerably. In the first stage there
is very little branching of the dendritic and
axonal processes. Hence volume of the fi-
brous mass is less than that of the cortex.
At each of the higher stadia extensive
branching of the dendrites and to a lesser
extent the axons, takes place. The small,
fine fibers which are considered sensory
terminals also increase in number.
GROWTH OF THE SPIDER NERVOUS SYSTEM
Axons from neurosecretory cells also ex-
hibit a similar growth pattern. But the most
conspicuous part of their growth is in form-
ing pools of stainable material. Such pools
increase during the peak period of secretory
activity and also from early to later stadia
(Babu, ’73); and there is an increase in the
extent of their ramification.
The only special neuropilar structure
present in the orbweb spider is the central
body. In the first instar a clearly recogniz-
able and demarkated central body is absent.
The central body becomes demarkated from
the general neuropile by mostly astral type
glial cells in the second stage, and further
differentiates into lobes towards the end of
the second stage. At each of the later stages
there is a corresponding increase in thick-
ness and length of the body. In many hemi-
metabolous insects (Panov, ’59) the central
body is present at the time of hatching. But
in holometabolous insects the first appear-
ance of the central body ranges from em-
bryonic stages as in Tenebrio, Antheraea,
and Culex (Panov, ’59; Hinke, ’61), through
larval stages, as in Danaus (Nordlander
and Edwards, ’68b) to the pupa as in Cal-
liphora (Gierying, ’65).
The differentiation of the central body
and the beginning of web construction have
been found to occur at the same time. In
the first and the earlier part of the second
stage of the spiderling only single threads
are formed. Towards the later part of the
second stadiumHwhen a demarkated cen-
tral body is formed, the spider begins to
construct its first small web with radii and
spirals. Thus a time correlation between
the formation of the central body and the
beginning of web construction was noticed.
Growth of neuroglial elements
The entire cephalothoracic nerve mass is
enveloped by a sheath called the neural
lamella. The number of layers in the con-
nective tissue normally varies from four to
ten. These layers widen at irregular inter-
vals to enclose a connective tissue cell nu-
cleus. The thickness of the sheath varies
considerably depending upon the location
and stadia. From the first to the sixth stage
of the spider, the thickness of the sheath
varies from 1 to 2 p all around the nerve
mass. It remains at this level on the mid
dorsal region of the supraesophageal gan-
335
glion throughout the life span of the spider.
From the seventh to the last stage the
sheath thickness gradually increased on
the ventral and dorsal region of the sub-
esophageal mass, reaching a maximum of
4 fj,. The greatest increase in thickness of
the neurilemmal sheath occurs at the origin
of major nerves. In the seventh stage the
sheath measured 8 n in the pedipalpal
and leg nerve region. In subsequent stages
it rose to 20^40 Maximal increase in
sheath thickness was noticed in gravid fe-
males, immediately after the eighth molt,
the sheath near the root of the pedipalpal
nerve measured 30 to 40 p. This increased
to 80—100 fji twenty days after the last
molt, at which time the animal is full of
eggs. Such differential growth of neural
lamella in adults is suggestive of an impor-
tant role in reproduction of the animal. In
some cases the sheath serves as a neuro-
hemal organ for storing and releasing the
neurosecretory products (Coggeshall, ’67;
Frazier et al., ’67; Rosenbluth, ’63; Simp-
son et al., ’66). Legendre (’59) suggested an
endocrine role for the neurilemma of
spiders.
In the seventh stage of a male spider the
thickness of the neural lamella varied from
0.5 fi to 4 /i. in different regions of the ce-
phalic nerve mass.
The neural lamella is not only found as
an outer covering of the nerve mass but
also as a separating sheath between the
fused ganglionic masses (fig. 12). Since the
fusion of ganglia is complete at the time
of hatching, these septal layers (fig. 12:
INL) can be distinguished in all instars.
The total number of these septal layers
corresponds to the total number of fused
ganglia.
The neural sheaths between the ganglia
also fuse together enclosing a small groove
or canal running in the dorso-ventral direc-
tion. In the groove and between the sheath
layers, granules of different diameters are
present. These are presumed to be nutritive
material on its way into the ganglion. Ac-
cording to Heywood (’65) such granules
form the permanent or semi-permanent
storage of food, not food in readily available
form. In mid sagittal sections where these
intra-ganglionic connective sheaths are
seen, it gives the appearance of blood ves-
sels running through the ganglion. Careful
336
K. SASIRA BABU
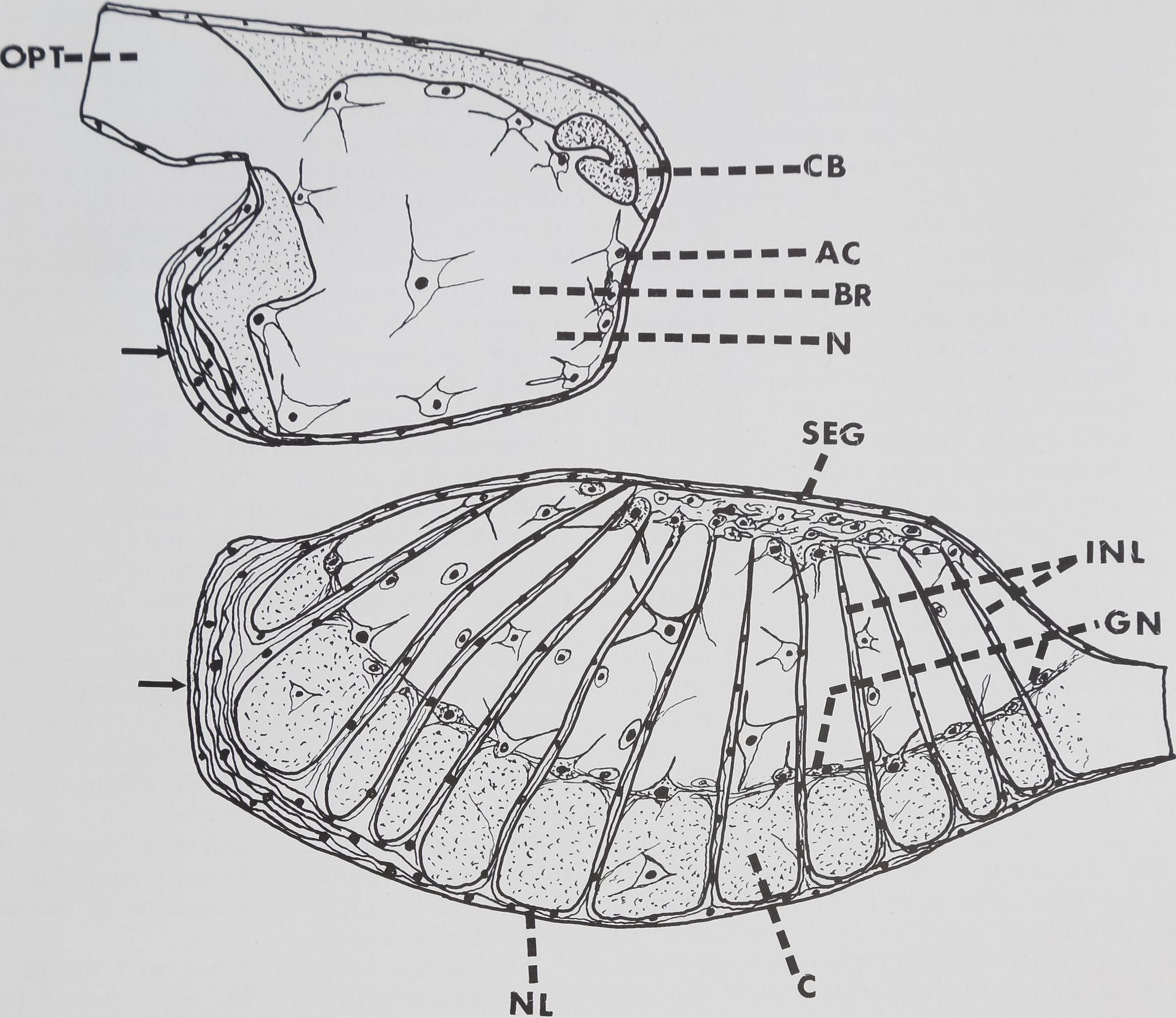
Fig. 12 Schematic drawing of cephalic nerve mass in mid sagittal section showing distribution of
neural lamella and glial cells in brain and subesophageal ganglion. The intraganglionic neural lamellae
represented, are only an approximate number. AC, Astral type of glial cells; BR, Brain; C, Cortex; CB,
Central body; GN, Glial cell zone between cortex and neuropile; INL, Intraganglionic neural lamellae;
N, Neuropile; NL, Neural lamellae; OPT, Optic nerve; SEG, Subesophageal ganglion. Arrows represent
thickened areas of neural lamellae.
observations of serial sections in the three
cardinal planes reveal that these are not
blood vessels. Further work is necessary
to confirm this observation by using special
techniques for tracing blood vessels. Until
then it is proposed that the cephalothoracic
nerve mass of spiders is avascular.
These intraganglionic sheaths have the
same characteristics as the outer neural
lamella. In both, the sheath layers run par-
allel and nuclei are found at irregular inter-
vals. The intra ganglionic sheaths are con-
tinuous with the inner layers of the outer
neural lamella. In the central or mid sag-
ittal region, the outer layers of the neural
lamella of the two adjunct ganglia are
tucked in so as to enclose the canal.
The outer neural lamella is chiefly a com-
pact sheath. But the layers in the periph-
eral region of the central canal are loosely
bound. These layers enter into the cortical
and fibrous areas at different levels and an
increase in their intrusion was noticed in
higher stadia. In the leech, Hirudo, pene-
tration of the neural lamella into central
ganglia has been reported (Coggeshall
and Fawcett, ’64). Thus the neural lamella
is not a mere outer investing sheath but it
invades deep into the cortical and central
fibrous zones.
Legendre (’59) described four types of
neuroglial cells in Tegenaria. Similar types
are noticed in the CNS of Argiope. The glial
cells, particularly of the astral type, are ar-
GROWTH OF THE SPIDER NERVOUS SYSTEM
ranged in such a way that they further di-
vide each ganglion into several zones in
female and male spiders.
This differentiation starts even in the
first instar and becomes more and more pro-
nounced in subsequent stages. A thick
zone of glial cell layer is formed between
the cortex and fibrous mass. On the dorsal
side of the subesophageal mass and near
the exit of major nerves, a similar concen-
tration of glial cells was noticed. It is diffi-
cult to identify them in the cortex but they
are relatively easy to identify in other
places. In the thicker zones, the astrocyte
type cells with denser cytoplasm are abun-
dant. In the fibrous mass, however, the
astrocyte type with clear cytoplasm is abun-
dant. The latter is found around the large
axons in the dorsal region of the neuropile
where motor axons are present. The size
of the glial cells shows little increase in
diameter from early to later stages. The
growth in a glial cell is mainly through ex-
tensions of ramifications. Unlike the nerve
cells the glial cells show an increase in
number during post embryonic develop-
ment. The astral type cells were counted in
the fibrous region because of easier identi-
fication. In the first instar the total astral
type glial cells are 2,574 ± 80. Their num-
ber increased to 6,580 ± 150 in the sixth
stage. In the post cocoon period the astral
cells increased to 22,040 ± 235. This shows
a tenfold increase in number of the astral
cells from early to last stage. The other
types of glial cells also increased in
number.
DISCUSSION
For comparison and evaluation, studies
like the present one, are lacking for most
of the invertebrate groups, except insects.
The developmental and anatomical organ-
ization significantly determines the integra-
tive functions of the nervous system. Hence,
knowledge of the growth pattern of neural
and non-neural elements in the CNS is es-
sential.
Volumetric changes in the growth of the
insect nervous system show a wide range
of patterns in brain growth (Edwards, ’69).
In Holometabola, the brain and whole body
growth has a negative allometric relation
in larval stages (Power, ’52; Hinke, ’61;
Nordlander and Edwards, ’68b). In hemi-
metabolous insects the growth relationship
between the brain or the ganglion and the
337
whole body bears a negative allometric re-
lation during post embryonic development
(Neder, ’59; Gymer and Edwards, ’67) as in
the spider Argiope aurantia.
Gymer and Edwards (’67) had reported
that the terminal ganglion of the house
cricket increased 40 fold during post em-
bryonic development. In Argiope the ce-
phalic nerve mass showed a 24 fold increase
in volume. Several component parts of the
CNS contribute to this growth pattern. In
the cricket an increase in cell volume rath-
er than cell number was reported. In the
spider the growth of the cortex is due to an
increase in cell size. This increase in Type-
B cells is 50% and in the largest Type-D
cells 600%. The large Type-D motor neu-
rons show further differentiation into dif-
ferent size categories where the number in
each category increases during post em-
bryonic development. Similar observations
were made in the brain of the beetle Popil-
lia which grows by increase in cell size
(Abercrombie, ’36). Monopolar neurons are
characteristic of invertebrate nervous sys-
tems, including spiders (Babu, ’69). The
soma, detached from dendritic and axonal
processes, is principally trophic in function
and does not participate in nerve conduc-
tion (Bullock and Horridge, !65). Thus the
enormous increase in cell volume and the
differential growth of motor cells is pre-
sumably due to the demand of growing
body organs and to an increase of dendritic
and axonal processes at later stadia of fe-
male spiders.
The growth of the cortex results also
from an increase in the number of glial
cells during development. Such glial cell
increase occurs amidst groups of neurons,
in the prominent glial zone between cortex
and neuropile and is due to increased in-
trusions of neural lamella into the cortex.
The increase in axon diameters within the
cortex also contributes to an increase in
volume.
Much of the neural growth, especially in
later stages is due to the growth of fibrous
mass. The cortical volume in the total nerve
mass exceeds the neuropile volume in
growth rate in the first two stages in Argi-
ope; in the first four instars of Acheta
(Gymer and Edwards, ’67) and in the pre-
pupal and pupal period of Drosophila (Pow-
er, ’52). In spiders the dendritic and axonic
processes of neurons, especially of type-D
cells grow extensively. The fiber diameter
K. SASIRA BABU
338
of large cells increases from 1 /x in the first
stage to a maximum of 16 /i in adults. As
the size category of cells increases, there is
a corresponding increase in size categories
of axons. The glial cell proliferation and
the increase in cell volume is enormous.
In Argiope one type of astral cells counted
increased ten fold.
Glial cells of all four types increase in
volume and number during post embryonic
development in Danaus (Nordlander and
Edwards, ’68b) and Pieris (Heywood, ’65).
In Acheta (Gymer and Edwards, ’67) the
number of glial cells increased from 3,400
to 20,000 during post embryonic develop-
ment. In adult spiders the astral type glial
cells with clear cytoplasm were observed to
extend their processes around the larger
axons which are presumably motor in the
dorsal region of the fibrous mass. Glial cells
are known to form sheaths around axons
in invertebrates Bin electron microscopic
studies, Edwards (’67) had shown in cereal
nerves of crickets that axons over 1 n diam-
eter have individual glial sheaths, and
larger axons have more elaborate glial
sheaths. Such relationships between glial
cells and axons were also reported in Aply-
sia (Batham, ’61; Coggeshall, ’67) and
Hirudo (Coggeshall and Fawcett,®64). In
the spider the neural lamellae also in-
crease enormously at the origin of main
nerves while remaining static at most other
places. At the intrusions of neural lamella
into the fibrous mass, the thickness of the
glial zone around the neuropile also in-
creases in post embryonic development.
The growth of neuropile is also depend-
ent upon incoming sensory fibers. At each
molt, new sensory neurons are differenti-
ated from ordinary ectodermal cells and
send their axons into the CNS where they
are incorporated into it (Wigglesworth, ’54).
The hair receptors of the abdominal cerci
of the house cricket increase in number
from 50 to 750 as the animal goes through
successive molts (Edwards, ’67). Similarly
there is an increase with age in the size and
the number of ommatidia in the eye of in-
sects (Bodenstein, ’53; Wigglesworth, ’65)
and of arachnids (Waterman, ’54) and a
concomitant increase in number of fibers
they send to the CNS. In the major meta-
thoracic nerve of the house cricket (Ed-
wards, ’67) the nerve growth is achieved by
addition of both sensory and motor fibers
and by an increase in the diameter of fibers.
The neural mass in Argiope is shown to
grow independent of the molting cycle. A
similar observation was made in Drosoph-
ila (Power, ’52) where the CNS grows
smoothly without showing any relationship
to molting. But Edwards (’69) suggests to
accept such conclusion with caution, since
glial cells show cyclic patterns of mitosis
in Acheta (Panov, ’61) and DNA synthesis
in Danaus (Nordlander and Edwards, ’68a).
In Argiope, the growth patterns of the
brain and of the subesophageal ganglion
show an inverse relationship. From the
first to the last stage in the female the
brain increases 10 fold, the subesophageal
mass 36 fold, and the céphalothorax 180
fold. The brain in the first stage constitutes
1/4 volume of the céphalothorax, and this
is reduced to l/90th in adult spiders. The
subesophageal mass on the other hand is
l/6th in the first stage, but occupies l/30th
volume of the céphalothorax in the adult
animal. The brain volume is relatively
greater in the first two stages, but in the
remaining stages the subesophageal vol-
ume is greater. The number of neuromeres
which fuse to form the brain is two: the
protocerebrum and tritocerebrum. But in
the subesophageal ganglion there are five
thoracic and approximately eleven abdom-
inal segments. Besides, except for a few
cells in the tritocerebrum, the rest are
small cells with fine processes, which show
a slow growth rate. Such neurons are con-
sidered as association fibers. The neuropile,
even though of diffuse type, is compactly
packed and also contains one specialized
mass, the central body. In the SEG there
is great diversity in cell size, and the cells
increase in volume during post embryonic
development. The larger cells are motor
cells with well developed dendritic arbori-
zations (Babu, ’69). The size of axons in
the neuropile also shows a similar growth
rate. Moreover, the neuropile is of the dif-
fuse type with loosely packed fibrous mat-
ter, and the compact special neuropilar
structures are absent. A casual observation
reveals that there are more glial cells and
specially thickened glial areas in the sub-
esophageal mass.
The morphology of the CNS (Babu, ’65,
’69) shows that the subesophageal gan-
glion is the major recipient of sensory input
from pedipalps, legs, céphalothorax and
abdomen. On the other hand, the brain re-
ceives sensory input primarily from eyes and
GROWTH OF THE SPIDER NERVOUS SYSTEM
339
chelicerae. In Argiope the optic centers are
poorly developed, while mushroom bodies,
olfactory and antennal centers of other
arthropods are lacking. These factors may
contribute to the growth differences of
brain and subesophageal mass.
Yet the breakdown of total number of
nerve cells to individual neuromers shows
that the protocerebrum has still the largest
number (approximately 13,000 in female
and 8,000 in male spiders) of cells. The
number of nerve cells for the other gan-
glion range from 2,000-750 in females and
males respectively. Due to reasons men-
tioned earlier and by virtue of the large
number of small chromatin rich cells, with
a specialized neuropilar mass like the cen-
tral body, the brain functions as an im-
portant integrating center.
Post embryonic changes in number of
neurons seem to vary in different inverte-
brate groups. In Argiope, the number of
nerve cells remained constant at 28, 853
± 1,655 for female and 21,648 ± 1,803
for male spiders. In the post embryonic
stages, division of nerve cells was not ob-
served with the stains employed. In hemi-
metabolous insects, Acheta, the number
of neurons in the last abdominal ganglion
remains relatively constant at 2,100 neu-
rons throughout development (Gymer and
Edwards, ’67). In the thoracic ganglion of
Oncopeltus a similar observation was made
by Johannson C57).
However, certain cell types in the brain
of insects increase in number during post
embryonic growth. In the optic lobes (Pan-
ov, ’63) and the Corpora peduncula (Ed-
wards, ’69) of insects the cell number
continues to increase throughout the post
embryonic life. In holometabolous insects a
large number of neurons is added during
post embryonic development and metamor-
phosis in the brain (Norlander and Ed-
wards, ’68a) and in thoracic ganglia (Hey-
wood, ’65). In the earthworm the number of
neurons increases in most parts of the brain
and this increase is particularly prominent
in ganglia which control the reproductive
apparatus (Ogawa, ’39). A similar differen-
tial increase in neuronal number was also
reported in Aplysia (Coggeshall, ’67; Fra-
zier et al., ’67). In the brain of Octopus
(Packard and Albergoni, ’70) estimates of
cell number based on their DNA content,
showed continuous increase from early to
adult stages.
A correlation was suggested between the
formation of the central body and begin-
ning of web construction in Argiope. Abla-
tion and stimulation of brain regions in
several arthropods were done successfully.
This has enabled investigators to demon-
strate a variety of behavioral activities in
bees (Vowles, ’61, ’64), locusts (Rowell,
’63), grasshoppers and crickets (Huber,
’67). In cricket and grasshopper (Huber,
’67), if the central body was destroyed or
hemisectioned, stridulation and associated
behaviour disappeared. On the other hand,
stimulation of the central body gave rise to
songs with some change in temporal pat-
terning. Thus the central body in insects is
an important coordinating and integrating
center.
The integrative functions of the nervous
system depend on its anatomy and on spe-
cific connections and the relationship of
cells. Anatomical studies in spider brain re-
veal that the central body is linked with
major pathways from the legs to the sub-
esophageal ganglion (Babu, ’65; Meier,
’67). Preliminary studies on the effects of
laser lesions on the central nervous system
were made (Witt, et al., ’64; LeGuelte and
Witt, ’68; Witt, ’69; LeGuelte and Witt,
’71). Lesions in the cortex close to the cen-
tral body produced disturbances in both
radii and spirals of the web. Further work
designed for ablation and with implanted
electrodes in the brain will help to localize
the functional organization of the CNS, es-
pecially with respect to web building. It was
reported that motor patterns of song pro-
duction and flight in the field cricket Tel-
eogryllus appear in a specific sequence
during the last four molts. Although neu-
ronal cell bodies may be present at hatch-
ing, neural circuits underlying adult motor
programs are not functional in early instars
(Bentley and Hoy, ’70). In spiders the be-
ginning of web building in the later part
of the second stage and cocoon spinning in
adult animals may involve similar neural
mechanisms developed during post embry-
onic growth.
Witt, Rawlings and Reed (’72) have re-
ported that the fine detail of the web un-
dergoes change throughout the life time of
the spider. New behavioral patterns like
spinning of the cocoon, absent in earlier
stages, will develop in adult spiders. The
causes for this new pattern of behavior
may be the development of reproductive or-
K. SASIRA BABU
g an s, The CNS of spiders increases in vol-
ume throughout ontogeny while maintain-
ing a constant number of nerve cells. It is
suggested that specific neural contacts in
the neuropile change so as to meet the new
demands of the growing body organs. Neu-
rons respond to normal events by meaning-
ful movements of axonal or dendritic ter-
minals (Bullock and Horridge, ’65). The
late emergence of motor patterns in crick-
ets was mentioned earlier. Presumably, in
the spider while sacrificing the detail in
web construction without much loss in
functional efficiency, new changes in the
nervous system occur in order to meet the
adult behavioral patterns.
The CNS of female and male spider
Argiope show certain interesting growth
patterns during post embryonic develop-
ment. Even though the body weight of male
and female spiders is equal in the fourth
and fifth stage, the growth slows down as
the male attains maturity. The male body
weighs one third of the female in the sev-
enth stage. This is largely because of poor
development of body musculature.
Volumetric measurements of the neural
mass and its component parts like cortex
and neuropile in male spiders are consist-
ently smaller than that of females through-
out. But a considerable difference between
male and female spider is noticed in cel-
lular organization. The total number of
nerve cells counted in males is smaller than
that of females by 11% in the SEG and
58% in the brain. The total number of
cells in the brain of males is 30% less than
in the SEG. In contrast the number of cells
in the brain of females is 13% more than
in the SEG. In the SEG, while three dif-
ferent size groups and a varied number of
motor neurons (325 cells of 20-29 p,; 30
cells of 30—39 p and 10 cells of 40—55 p)
are noticed in the seventh stage of the fe-
male, only 63 cells of the small size group
are present in mature males.
If animal behavior is “What an animal
does,” then development is an aspect of be-
havior (Edwards, ’67). The anatomical dif-
ferences between male and female spiders
during development of the CNS may partly
explain the behavioral peculiarities of both
sexes.
ACKNOWLEDGMENT
I wish to express my gratitude to Dr.
Peter N. Witt for his constant interest and
suggestions during the course of this work.
My thanks are also due to Prof. David E.
Davis for his interest and to Dr. Frank
Enders in rearing animals. The help of
Mr. P. Murali Mohan, D. Venkateswarlu
and T. Pavan Kumar is acknowledged. This
work has been carried out in the Research
Division of the North Carolina Department
of Mental Health and Department of Zool-
ogy, North Carolina State University, Ral-
eigh, during the tenure of a senior NSF
fellowship to the author, and with partial
support from the North Carolina Depart-
ment of Mental Health and NSF grant GB-
6246 to Dr. Peter N. Witt.
LITERATURE CITED
Abercrombie, W. F. 1936 Studies on cell num-
ber and the progression factors in the growth of
Japanese beetle larvae (Popillia japonica New-
man). J. Morph., 59: 91—112.
Babu, K. S. 1965 Anatomy of the central nervous
system of Arachnids. Zool. Jb., Anat., 82: 1—154.
——– 1969 Certain histological and anatomi-
cal features of the central nervous system of a
large Indian spider, Poecilotheria. Am. Zoolo-
gist, 9: 113-119.
——– 1973 Histology of the neurosecretory sys-
tem and neurohaemal organs of the spider, Argi-
ope aurantia (Lucas). J. Morph., 141(1): 77—97.
Batham, E. J. 1961 Infolding of nerve fibre mem-
branes in the ophisthobranch mollusc, Aplysia
Californie a. J. Biophys. Biochem. Cytol., 9: 490—
492.
Bentley, R. D., and R. R. Hoy 1970 Postembryon-
ic development of adult motor patterns in crick-
ets: A neural analysis. Science, 170: 1409—1411.
Blanchard, E. 1859 L’organisation du Règne ani-
mal. Arachnides. Paris, Victor Masson.
Bodenstein, D. 1953 Regeneration. In: Insect
Physiology. K. D. Roeder, ed. Wiley, New York.
Bullock, T. H., and G. A. Horridge 1965 Struc-
ture and Function in the Nervous System of In-
vertebrates. Freeman, San Francisco.
Buxton, B. H. 1917 Notes on the anatomy of
arachnids. J. Morph., 29: 13—31.
Coggeshall, R. E. 1967 A light and electron mi-
croscope study of the abdominal ganglion of
Aplysia californica. J. Neurophysiol., 30: 1263—
1287.
Coggeshall, R. E., and D. W. Fawcett 1964 The
fine structure of the central nervous system of the
leech, Hirudo medicinalis. J. Neurophysiol., 27:
229-289.
Edwards, J. S. 1967 Some questions for the in-
sect nervous system. In: Insects and Physiology.
J. W. L. Beament and J. E. Treherne, eds. Oli-
ver and Boyd, Edinburgh, pp. 163—173.
——- 1969 Postembryonic development and
regeneration of the insect nervous system. Ad-
van. Insect Physiol., 6: 97—137.
Firstman, B. 1954 The central nervous system,
musculature and segmentation of the cephalo-
GROWTH OF THE SPIDER NERVOUS SYSTEM
341
thorax of a Tarantula (Eurypelma californiens,
Ausserer) (Arachnid). Mieroent., 19: 14—22.
Frazier, W. T., E. R. Kandel, I. Kupfermann, R.
Waziri and R. E. Coggeshall 1967 Morpholog-
ical and functional properties of identified cells
in the abdominal ganglion of Aplysia Califor-
nia. J. Neurophysiol., 30: 1288—1351.
Gerhardt, U., and A. Kaestner 1937 Araneae.
In: Kükenthal, Krumbach’s Handbuch der Zool-
ogie, 32: 418-427.
Gierying, R. 1965 Verànderungen der histolo-
gischen Struktur de Gehirns von Calliphora vom-
itoria (L.) (Diptera) wàhrend der postembry-
onalen Entwicklung. Z. wiss. Zool., 171: 80—96.
Gymer, A., and J. S. Edwards 1967 The develop-
ment of the insect nervous system. I. An analy-
sis of postembryonic growth in the terminal gan-
glion of Acheta domesticus. J. Morph., 123 : 191—
197.
Haller, B. 1912 fiber das Zen train erven system
der Skorpione und der Spinnen. Ein zweiter
Beitrag zur Stammesgeschichte der Arachnoid en.
Arch. Mikr. Anat., 79: 504—524.
Hanstrôm, B. 1919 Zur Kenntnfs des Zentralen
Nervensystems der Arachnid en and Pantapoden.
Inaug. Dissert., pp. 1-191.
——- 1921 fiber die Histologie und verglei-
chende Anatomie der Sehganglien und Globuli
der Araneen. Kgl. Svenska vet. Akad. Handb.,
61: 1-39.
——- 1928 Vergleichende Anatomie des Ner-
vensystems der wirbellosen Tiere. pp. 1—628.
——- 1935 Fortgesetzte Untersuchungen iiber
das Araneengehirn. Zool. Jb. Anat., 59: 455-478.
Heywood, R. B. 1965 Changes occurring in the
central nervous system of Pieris brassicaeWSM
(Lepidoptera) during metamorphosis. jHlnsect
Physiol., 11: 413^30.
Hilton, W. S. 1912 A preliminary study of the
central nervous system of spiders. J. Ent. Zool.,
4: 832-836.
Hinke, W. 1961 Das relative postembryon ale
Wachstum der Hirnteile von Culex pipiens.
Drosophila melanogaster und Drosophila mu-
tanten. Z. Morphol. Oekol. Tiere, 50: 81—118.
Huber, F. 1967 Central control of movements
and behaviour of invertebrates. In: Inverte-
brates. C. A. G. Wiersma, ed. University of Chi-
cago Press.
Johannson, A. S. 1957 The nervous system of the
milkweed bug, Oncopeltus fasciatus (Dallas)
(Heteroptera Lygaeidae). Trans. Am. ent. Soc.,
83:119-183.
Legendre, R. 1953 Le système sympathique sto-
matogastrique (“organe de Schneider”) des
araignées du genre Tegenaria. C. R. Acad. Sci.,
237:1283-1285.
1959 Contributions to a study of the ner-
vous system of Araneids (Contribution a l’étude
du système nerveux des aranéides). Ann. Biol.,
34: 193-223.
LeGuelte, L., and P. N. Witt 1968 Données mor-
phologiques permettant de localiser sur l’animal
vivant les differéntes régions du système nerveux
central de l’Araignée argiopide Araneus diade-
matus Cl. soumise à des lésions par action du
laser. Bull. Mus. Nat. Hist. Nat., 40: 742—744.
1971 Consequences histologiques et com-
portemantales de lesions (Par Laser) du ganglion
sus-oesophagien de l’araignée (Araneus diade-
matus Cl.). Rev. comp. Animal. T.5-Ler Trim,
pp. 19-26.
Levi, H. 1968 The spider genera Gea and Argiope
in America (Araneae: Araneidae). Bull. Mus.
Comp. Zool., 136: 319—352.
Marrable, A. W. 1962 The counting of cells and
nuclei in microtome sections. Quart. J. Micr.
Sci., 103: 331-347.
Meier, F. 1967 Beitrâge zur Kenntnis der post-
embryonal en Entwicklung der Spinnen Araneida,
Labidognatha, unter besonderer Berücksichti-
gung der Histogenèse des Z en train erven system s.
Rev. Suisse Zool., 74(1): 127.
Millot, J. 1949 Chelicérates., in Grassé’s: Traité
de Zoologie, 6: 263—743.
Neder, R. 1959 Allometrisches Wachstum von
Hirnteilen bei drei verscheiden grossen Schaben-
arten. Zool. Jb. allg. Zool. Physiol., 77: 411^164.
Nordlander, R. H., and J. S. Edwards 1968a
Morphological cell death in the postembryonic
development of the insect optic lobes. Nature
Jptond.), 218: 780-781.
——– 1968b Morphology of the larval and adult
brains of the Monarch butterfly Danaus plexippus
Wm$. Morph., 126: 67-94.
Ogawa, F. 1939 The nervous system of earth-
worm (Pheritima communissima) in different
ages. Sci. Rept. ResSnst. Tohoku UnivHB, 13:
395-488.
Packard, A., and V. Albergoni 1970 Relative
growth, nucleic acid content and cell numbers of
the brain in Octopus vulgaris (Lamarck). J.
exp. Biol., 52: 539—552.
Palmgren, A. 1948 A rapid method for selective
silver staining of nerve fibers and nerve endings
in mounted paraffin sections. Acta Zoologica,
29: 377-392.
Panov, A. A. 1959 Structure of the insect brain
at successive stages of postembryonic develop-
ment. II. The central body. Ent. Obozr., 38: 276—
284.
——— 1961 a The structure of the insect brain at
successive stages of postembryonic development.
IV. The olfactory center. Ent. Obozr., 40: 259—
271.
——— 1963 The origin and fate of neuroblasts,
neurons and neuroglial cells in the central ner-
vous system of the Chinese silkmoth, Antheraea
pernyi Guer (Lepidoptera, Attacidae). Ent.
Obozr., 42: 337—350.
Pocock, R. J. 1902 On some points in the ana-
tomy of the alimentary canal and nervous sys-
tem of the arachnidian suborder Pedipalpi. Proc.
Zool. Soc. Lond., 11: 169-172.
Power, M. E. 1952 A quantitative study of the
growth of the central nervous system of a holo-
metabolus insect, Drosophila melanogaster. J.
Morph., 91: 389-411.
Rosenbluth, J. 1963 The visceral ganglion of
Aplysia californica. Z. Zellforsch, 60: 213—236.
Rowell, C. H. F. 1963 A method for chronically
implanting stimulating electrodes into the brains
of locusts, and some results of stimulation. J.
exp. Biol., 40: 271-284.
Saint-Rémy, M. 1887 Contribution a l’étude du
cerveau chez les Arthropodes trachéates. Arch.
Zool. Exper., 5(2): 279.
Simpson, L., H.A. Bern and R. S. Nishioka 1966
Survey of evidence for neurosecretion in gastro-
pod molluscs. Am. Zoologist., 6: 123—138.
K. SASIRA BABU
Vowles, D. M. 1961 The physiology of insect ner-
vous system, Int. Rev. Neurobiol., 3: 349—373.
1964 Models and the insect brain. In:
Neural theory and modelling. R. F. Reiss, ed.
Stanford University Press, pp. 377—399.
Waterman, T. H. 1954 Relative growth and the
compound eye in Xiphosura. J. MorphoL, 95:
125-158.
Wigglesworth, V. B. 1954 The histology of the
nervous system of an insect, Rhodnius prolixins*
(Hemiptera). II. The Central ganglion. Quart. H
Micr. Sci., 100: 299-313.
—-—• 1965 The Principles of Insect Physiology.
Methuen, London.
Witt, P. N. 1969 Behavioral consequences of
Laser lesions in the central nervous system of
Araneus diadematus Cl. Am. Zoologist., 9: 121—
131.
——– 1971 Instructions for working with web-
building spiders in the laboratory. Bioscience,
21 (M 23-25.
Witt, P. N., J. D. Rawlings and C. F. Reed 1972
Ontogeny Of web-building behavior in two
orb-weaving spiders. Am. Zoologist, 12: 445—
454.
Witt, P. N., C. F. Reed and M K. Tittel 1964 La-
ser lesions and spider web construction. Nature,
20lBl50-152.