Warning: Trying to access array offset on value of type null in /home/elisew5/public_html/drpeterwitt.com/wp-content/plugins/contextual-related-posts/includes/content.php on line 49
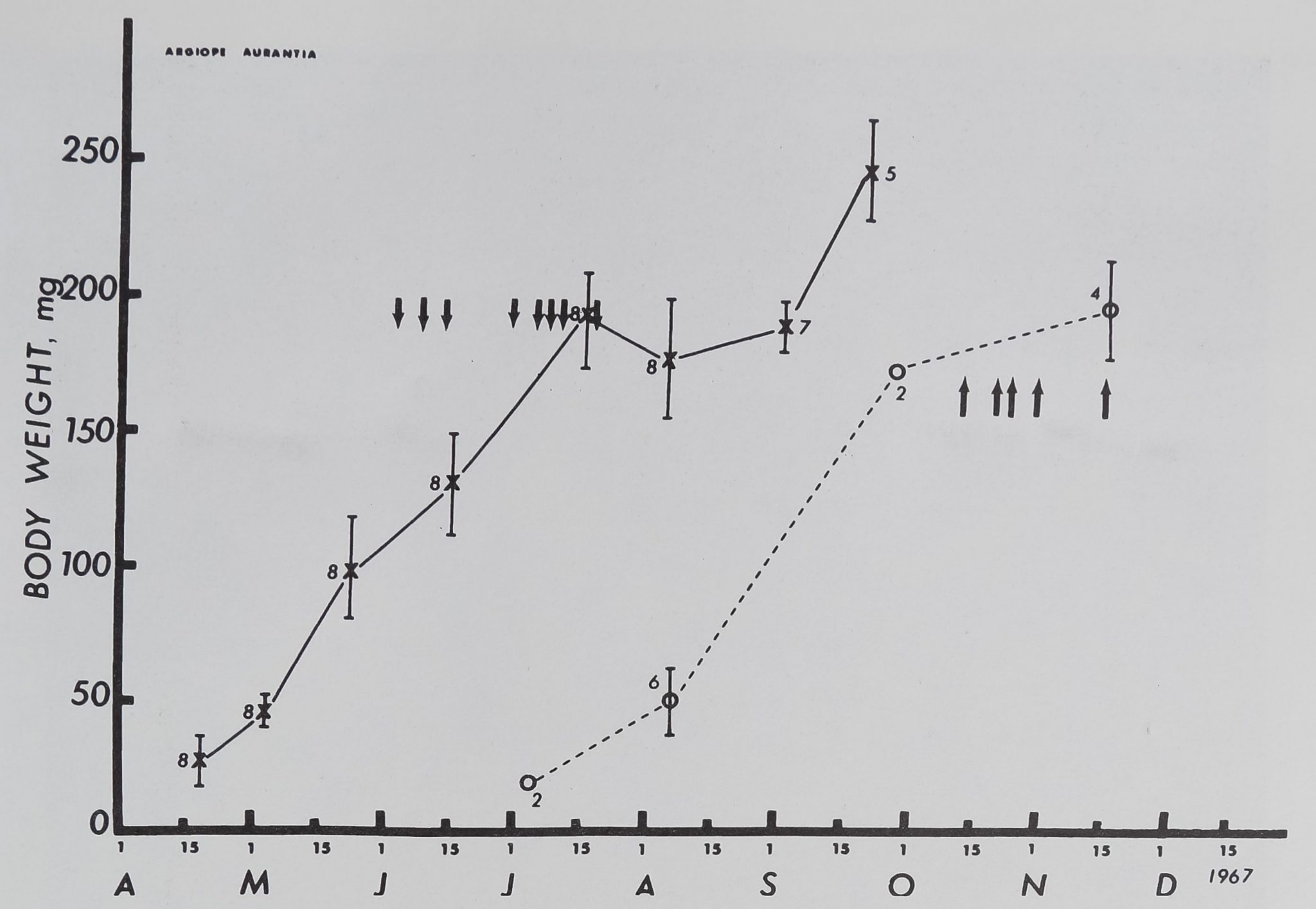
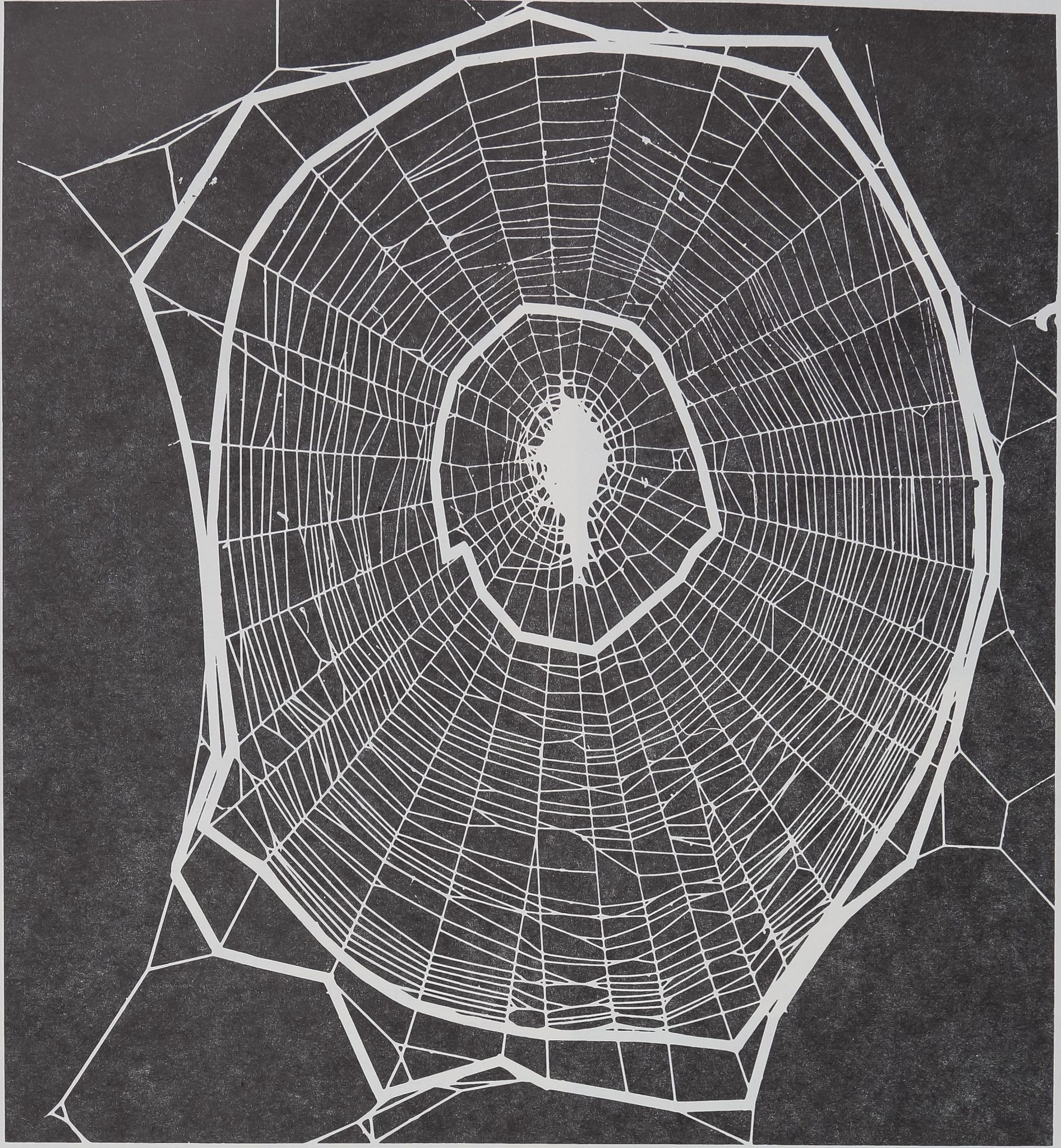
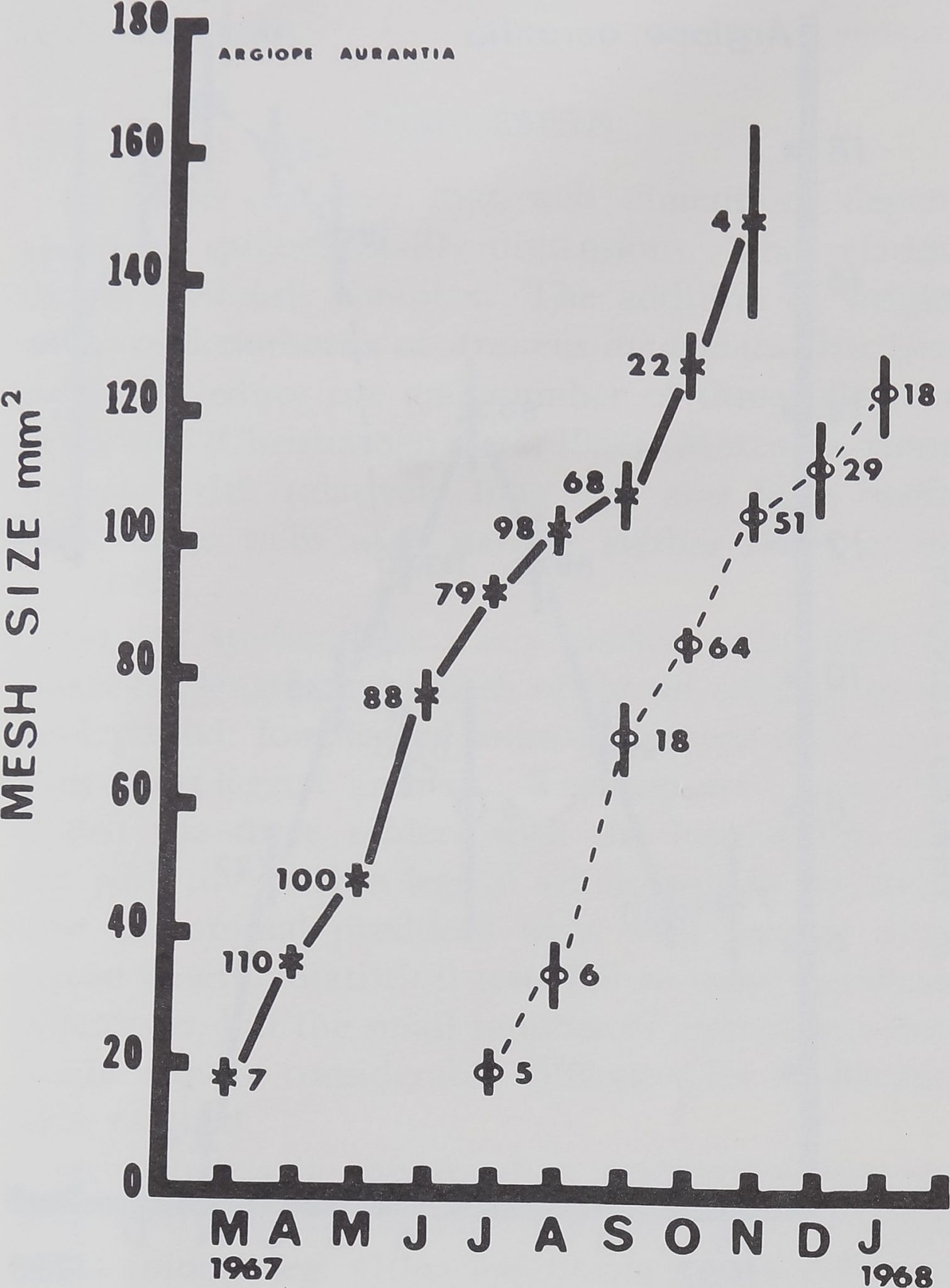
The Orb Web during the Life of Argiupe aurantia
Developmental Psychobiology
Warning: Trying to access array offset on value of type null in /home/elisew5/public_html/drpeterwitt.com/wp-content/plugins/contextual-related-posts/includes/content.php on line 49